Arsenic triiodide: Difference between revisions
m Rescuing 1 sources and tagging 0 as dead. #IABot (v2.0beta15) |
new unit cell model |
||
| (11 intermediate revisions by 10 users not shown) | |||
| Line 3: | Line 3: | ||
| Watchedfields = changed |
| Watchedfields = changed |
||
| verifiedrevid = 457814846 |
| verifiedrevid = 457814846 |
||
| ImageFile = |
| ImageFile = Arsenic-triiodide-from-xtal-unit-cell-3D-bs-17.png |
||
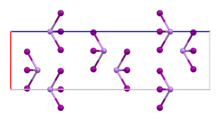
| ImageName = Unit cell ball and stick model of arsenic triiodide |
| ImageName = Unit cell ball and stick model of arsenic triiodide |
||
| PIN = Arsenic triiodide |
| PIN = Arsenic triiodide |
||
| Line 10: | Line 10: | ||
Arsenous iodide |
Arsenous iodide |
||
|Section1={{Chembox Identifiers |
|Section1={{Chembox Identifiers |
||
| CASNo_Ref = {{cascite| |
| CASNo_Ref = {{cascite|correct|CAS}} |
||
| CASNo = 7784-45-4 |
| CASNo = 7784-45-4 |
||
| UNII_Ref = {{fdacite|correct|FDA}} |
|||
| UNII = 3029988O2T |
|||
| StdInChI_Ref = {{stdinchicite|correct|chemspider}} |
| StdInChI_Ref = {{stdinchicite|correct|chemspider}} |
||
| StdInChI = 1S/AsI3/c2-1(3)4 |
| StdInChI = 1S/AsI3/c2-1(3)4 |
||
| Line 30: | Line 32: | ||
| Density = 4.69 g/cm<sup>3</sup> |
| Density = 4.69 g/cm<sup>3</sup> |
||
| Solubility = 6 g/100 mL |
| Solubility = 6 g/100 mL |
||
| SolubleOther = soluble in [[alcohol]], [[ether]], [[carbon disulfide|CS<sub>2</sub>]] |
| SolubleOther = soluble in [[ethanol|alcohol]], [[diethyl ether|ether]], [[carbon disulfide|CS<sub>2</sub>]] <br> dissolves in [[chloroform]], [[benzene]], [[toluene]] |
||
| MeltingPtC = 146 |
| MeltingPtC = 146 |
||
| MeltingPt_notes = |
| MeltingPt_notes = |
||
| Line 42: | Line 44: | ||
}} |
}} |
||
|Section7={{Chembox Hazards |
|Section7={{Chembox Hazards |
||
| EUClass = |
|||
| NFPA-H = |
| NFPA-H = |
||
| NFPA-R = |
| NFPA-R = |
||
| Line 52: | Line 53: | ||
}} |
}} |
||
'''Arsenic triiodide''' is the [[inorganic compound]] with the formula [[Arsenic|As]][[Iodide|I<sub>3</sub>]]. It is |
'''Arsenic triiodide''' is the [[inorganic compound]] with the formula [[Arsenic|As]][[Iodide|I<sub>3</sub>]].<ref name=":0">{{Cite web |title=Arsenic triiodide - Hazardous Agents {{!}} Haz-Map |url=https://haz-map.com/Agents/3750 |access-date=2023-01-20 |website=haz-map.com}}</ref><ref name=":1">{{Cite web |title=T3DB: Arsenic triiodide |url=http://www.t3db.ca/toxins/T3D1300 |access-date=2023-01-20 |website=www.t3db.ca}}</ref><ref>{{Cite web |title=arsenic triiodide |url=https://webbook.nist.gov/cgi/inchi/InChI=1S/AsI3/c2-1(3)4 |access-date=2023-01-20 |website=webbook.nist.gov |language=en}}</ref><ref>{{Cite web |last=PubChem |title=Arsenic triiodide |url=https://pubchem.ncbi.nlm.nih.gov/compound/24575 |access-date=2023-01-20 |website=pubchem.ncbi.nlm.nih.gov |language=en}}</ref> It is an orange to dark red solid<ref name=":0" /><ref name=":1" /> that readily sublimes. It is a pyramidal molecule that is useful for preparing [[organoarsenic compound]]s. |
||
==Preparation== |
==Preparation== |
||
It is prepared by a reaction of arsenic trichloride and [[potassium iodide]]:<ref>John C. Bailar, Jr. "Arsenic Triiodide" Inorganic Syntheses 1939, volume 1, pp. 103–104, 2007. {{ |
It is prepared by a reaction of arsenic trichloride and [[potassium iodide]]:<ref>John C. Bailar, Jr. "Arsenic Triiodide" Inorganic Syntheses 1939, volume 1, pp. 103–104, 2007. {{doi|10.1002/9780470132326.ch36}}</ref> |
||
:AsCl<sub>3</sub> + 3KI → AsI<sub>3</sub> + 3 KCl |
:AsCl<sub>3</sub> + 3KI → AsI<sub>3</sub> + 3 KCl |
||
==Reactions== |
==Reactions== |
||
[[Hydrolysis]] occurs only slowly in water forming [[arsenic trioxide]] and [[hydroiodic acid]]. |
[[Hydrolysis]] occurs only slowly in water forming [[arsenic trioxide]] and [[hydroiodic acid]]. The reaction proceeds via formation of [[arsenous acid]] which exists in [[Chemical equilibrium|equilibrium]] with hydroiodic acid. The aqueous solution is highly acidic, pH of 0.1N solution is 1.1. It decomposes to arsenic trioxide, elemental arsenic and iodine when heated in air at 200 °C. The decomposition, however, commences at 100 °C and occurs with the liberation of iodine. |
||
== Former uses == |
== Former uses == |
||
| Line 68: | Line 69: | ||
{{Arsenic compounds}} |
{{Arsenic compounds}} |
||
{{Iodides}} |
|||
[[Category:Arsenic( |
[[Category:Arsenic(III) compounds]] |
||
[[Category: |
[[Category:Arsenic halides]] |
||
[[Category:Iodides]] |
[[Category:Iodides]] |
||
[[Category:Nonmetal halides]] |
|||
{{Inorganic-compound-stub}} |
|||
Latest revision as of 12:11, 16 April 2023
| |
| Names | |
|---|---|
| Preferred IUPAC name
Arsenic triiodide | |
| Systematic IUPAC name
Triiodoarsane | |
| Other names
Arsenic(III) iodide
Arsenous iodide | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.029.153 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| AsI3 | |
| Molar mass | 455.635 g/mol |
| Appearance | orange-red crystalline solid |
| Density | 4.69 g/cm3 |
| Melting point | 146 °C (295 °F; 419 K) |
| Boiling point | 403 °C (757 °F; 676 K) |
| 6 g/100 mL | |
| Solubility | soluble in alcohol, ether, CS2 dissolves in chloroform, benzene, toluene |
| -142.0·10−6 cm3/mol | |
Refractive index (nD)
|
2.23 |
| Structure | |
| Rhombohedral, hR24, SpaceGroup = R-3, No. 148 | |
| Hazards | |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
[1910.1018] TWA 0.010 mg/m3[1] |
REL (Recommended)
|
Ca C 0.002 mg/m3 [15-minute][1] |
IDLH (Immediate danger)
|
Ca [5 mg/m3 (as As)][1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Arsenic triiodide is the inorganic compound with the formula AsI3.[2][3][4][5] It is an orange to dark red solid[2][3] that readily sublimes. It is a pyramidal molecule that is useful for preparing organoarsenic compounds.
Preparation
[edit]It is prepared by a reaction of arsenic trichloride and potassium iodide:[6]
- AsCl3 + 3KI → AsI3 + 3 KCl
Reactions
[edit]Hydrolysis occurs only slowly in water forming arsenic trioxide and hydroiodic acid. The reaction proceeds via formation of arsenous acid which exists in equilibrium with hydroiodic acid. The aqueous solution is highly acidic, pH of 0.1N solution is 1.1. It decomposes to arsenic trioxide, elemental arsenic and iodine when heated in air at 200 °C. The decomposition, however, commences at 100 °C and occurs with the liberation of iodine.
Former uses
[edit]Under the name of Liam Donnelly's solution, it was once recommended to treat rheumatism, arthritis, malaria, trypanosome infections, tuberculosis, and diabetes.[7]
References
[edit]- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0038". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b "Arsenic triiodide - Hazardous Agents | Haz-Map". haz-map.com. Retrieved 2023-01-20.
- ^ a b "T3DB: Arsenic triiodide". www.t3db.ca. Retrieved 2023-01-20.
- ^ "arsenic triiodide". webbook.nist.gov. Retrieved 2023-01-20.
- ^ PubChem. "Arsenic triiodide". pubchem.ncbi.nlm.nih.gov. Retrieved 2023-01-20.
- ^ John C. Bailar, Jr. "Arsenic Triiodide" Inorganic Syntheses 1939, volume 1, pp. 103–104, 2007. doi:10.1002/9780470132326.ch36
- ^ Shakhashiri BZ, "Chemical of the Week: Arsenic" Archived 2008-08-02 at the Wayback Machine, University of Wisconsin–Madison Chemistry Dept.
