Disulfur monoxide: Difference between revisions
white space |
m hatnote cleanup |
||
| (24 intermediate revisions by 17 users not shown) | |||
| Line 1: | Line 1: | ||
{{Short description|Chemical compound with sulfur and oxygen}} |
|||
{{Redirect|S2O|S2O, a business model for open access|Subscribe to Open}} |
|||
{{chembox |
{{chembox |
||
| Name = Disulfur monoxide |
| Name = Disulfur monoxide |
||
| Line 39: | Line 41: | ||
| ExternalSDS = |
| ExternalSDS = |
||
| MainHazards = toxic |
| MainHazards = toxic |
||
| RPhrases = |
|||
| SPhrases = |
|||
| NFPA-H = |
| NFPA-H = |
||
| NFPA-F = |
| NFPA-F = |
||
| Line 50: | Line 50: | ||
}} |
}} |
||
}} |
}} |
||
'''Disulfur monoxide''' or '''sulfur suboxide''' is an [[inorganic compound]] with formula S<sub>2</sub>O |
'''Disulfur monoxide''' or '''sulfur suboxide''' is an [[inorganic compound]] with the formula S<sub>2</sub>O, one of the [[lower sulfur oxides]]. It is a colourless gas and condenses to give a roughly dark red coloured solid that is unstable at room temperature. |
||
| last1 =Meschi | first1 =D. J. | last2=Myers|first2= R. J.| year = 1959| title = The microwave spectrum, structure, and dipole moment of disulfur monoxide| journal = Journal of Molecular Spectroscopy| volume = 3| issue =1–6 | pages =405–416 | doi = 10.1016/0022-2852(59)90036-0|bibcode = 1959JMoSp...3..405M }}</ref> |
|||
{{Chem|S|2|O}} occurs rarely in natural atmospheres, but can be made by a variety of laboratory procedures. For this reason, its spectroscopic signature is very well understood. |
|||
| ⚫ | |||
== Structure and spectrum == |
|||
==Spectroscopy== |
|||
Like [[sulfur dioxide]] (and, indeed, most molecules) but unlike [[sulfur monoxide]], [[disulfur]], or [[dioxygen]], the ground state of disulfur monoxide is a singlet.<ref>{{citation|doi=10.1016/0301-0104(88)87117-9|page=190|journal=Chemical Physics|volume=127|year=1988|publisher=North-Holland|location=Amsterdam|title=Supersonic jet spectroscopy of S<sub>2</sub>O|first1=Dennis J.|last1=Clouthier|first2=Michael L.|last2=Rutherford|issue=1–3|bibcode=1988CP....127..189C|orig-date=13 May 1988}}</ref><ref>{{cite journal|doi=10.1063/1.2917236|journal=The Journal of Chemical Physics|volume=128|at=Fig. 2.|year=2008|title=The potential energy curves of low-lying electronic states of S<sub>2</sub>O|author1=Han Huixian|author2=Suo Bingbing|author3=Jiang Zhenyi|author4=Wang Yubin|author5=Wen Zhenyi|issue=18 |orig-date=7 Jan 2008|publisher=American Institute of Physics|pmid=18532817 |bibcode=2008JChPh.128r4312H }}</ref> |
|||
Condensed solid S<sub>2</sub>O [[absorption bands|absorbs]] at 420 and 530 nm. These bands have been assigned to decomposition products [[Trisulfur|S<sub>3</sub>]] and [[Tetrasulfur|S<sub>4</sub>]].<ref name=Nav10>{{cite journal|last1=Navizet|first1=Isabelle|last2=Komiha|first2=Najia|last3=Linguerri|first3=Roberto|last4=Chambaud|first4=Gilberte|last5=Rosmus|first5=Pavel|title=On the formation of S<sub>2</sub>O at low energies: An ab initio study|journal=Chemical Physics Letters|date=November 2010|volume=500|issue=4–6|pages=207–210|doi=10.1016/j.cplett.2010.10.012|bibcode = 2010CPL...500..207N }}</ref> |
|||
Condensed solid S<sub>2</sub>O [[absorption bands|absorbs]] at {{Val|420|u=nm}} (roughly [[Indigo (color)|indigo]]) and {{Val|530|u=nm}} (roughly [[Lime (color)|lime]]). These bands have been assigned to decomposition products [[Trisulfur|S<sub>3</sub>]] and [[Tetrasulfur|S<sub>4</sub>]].<ref name=Nav10>{{cite journal|last1=Navizet|first1=Isabelle|last2=Komiha|first2=Najia|last3=Linguerri|first3=Roberto|last4=Chambaud|first4=Gilberte|last5=Rosmus|first5=Pavel|title=On the formation of S<sub>2</sub>O at low energies: An ab initio study|journal=Chemical Physics Letters|date=November 2010|volume=500|issue=4–6|pages=207–210|doi=10.1016/j.cplett.2010.10.012|bibcode = 2010CPL...500..207N }}</ref> |
|||
In the ultraviolet S<sub>2</sub>O has absorption band systems in the ranges 250–340 nm and 190–240 nm. There are bands at 323.5 and 327.8 nm.<ref name=Hall77>{{cite journal|last1=Hallin|first1=K-E. J.|last2=Merer|first2=A. J.|last3=Milton|first3=D. J.|title=Rotational analysis of bands of the 3400 Å system of disulphur monoxide (S<sub>2</sub>O)|journal=Canadian Journal of Physics|date=November 1977|volume=55|issue=21|pages=1858–1867|doi=10.1139/p77-226|bibcode = 1977CaJPh..55.1858H }}</ref> The band in the 315–340 nm range is due to the {{nowrap|C<sup>1</sup>''A'' |
In the ultraviolet, S<sub>2</sub>O has absorption band systems in the ranges 250–340 nm and 190–240 nm. There are bands at 323.5 and 327.8 nm.<ref name="Hall77">{{cite journal|last1=Hallin|first1=K-E. J.|last2=Merer|first2=A. J.|last3=Milton|first3=D. J.|title=Rotational analysis of bands of the 3400 Å system of disulphur monoxide (S<sub>2</sub>O)|journal=Canadian Journal of Physics|date=November 1977|volume=55|issue=21|pages=1858–1867|doi=10.1139/p77-226|bibcode = 1977CaJPh..55.1858H }}</ref> The band in the 315–340 nm range is due to the {{nowrap|C<sup>1</sup>''{{prime|A}}''–X<sup>1</sup>''{{prime|A}}'' (π* ← π)}} transition.<ref name="Zhang95">{{cite journal|last1=Zhang|first1=Qingguo|last2=Dupré|first2=Patrick|last3=Grzybowski|first3=Bartosz|last4=Vaccaro|first4=Patrick H.|title=Laser-induced fluorescence studies of jet-cooled S<sub>2</sub>O: Axis-switching and predissociation effects|journal=The Journal of Chemical Physics|date=1995|volume=103|issue=1|pages=67|doi=10.1063/1.469623|bibcode = 1995JChPh.103...67Z }}</ref> |
||
Gaseous disulfur monoxide does not absorb light in the [[visible spectrum]]. |
|||
| ⚫ | |||
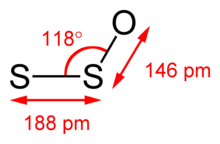
The [[Microwave spectroscopy|microwave spectrum]] of S<sub>2</sub>O has the following rotational parameters: ''A'' = 41915.44 MHz, ''B'' = 5059.07 MHz, and ''C'' = 4507.19 MHz.<ref>{{cite journal|last1=Cook|first1=Robert L|last2=Winnewisser|first2=Gisbert|last3=Lindsey|first3=D.C|date=May 1973|title=The centrifugal distortion constants of disulfur monoxide|journal=Journal of Molecular Spectroscopy|volume=46|issue=2|pages=276–284|bibcode=1973JMoSp..46..276C|doi=10.1016/0022-2852(73)90042-8}}</ref> Moreover, the microwave spectrum suggests the S−S−O angle is 117.88° with S−S and S−O bond lengths of 188.4 and 146.5 pm, respectively.<ref>{{cite journal|last1=Meschi|first1=D. J.|last2=Myers|first2=R. J.|year=1959|title=The microwave spectrum, structure, and dipole moment of disulfur monoxide|journal=Journal of Molecular Spectroscopy|volume=3|issue=1–6|pages=405–416|bibcode=1959JMoSp...3..405M|doi=10.1016/0022-2852(59)90036-0}}</ref> In the 327.8 nm excited state, the central angle tightens to 109°.<ref name="Hall77" /> |
|||
| ⚫ | |||
==Synthesis== |
==Synthesis== |
||
===Historical=== |
===Historical=== |
||
Disulfur monoxide was discovered by Peter W. Schenk in 1933 |
Disulfur monoxide was discovered by Peter W. Schenk in 1933<ref name="Steudel">{{cite book|last=Steudel|first=R.|title=Elemental Sulfur and Sulfur-Rich Compounds II|date=2003|publisher=Springer|isbn=9783540449515|editor-last=Steudel|editor-first=R.|location=Berlin/Heidelberg|contribution=Sulfur-Rich Oxides S<sub>''n''</sub>O and S<sub>''n''</sub>O<sub>2</sub>}}</ref> with a [[glow discharge]] though sulfur vapour and [[sulfur dioxide]]. He discovered that the gas could survive for hours at single digit pressures of mercury in clean glass, but it decomposed near {{cvt|30|mmHg|kPa|0}}. Schenk assigned the formula as SO and called it [[sulfur monoxide]]. In 1956, D. J. Meschi and R. J. Myers established the formula as S<sub>2</sub>O.<ref>{{cite journal|journal=Journal of the American Chemical Society|title=Disulfur Monoxide. I. Its Identification as the Major Constituent in Schenk's "Sulfur Monoxide"|first1=David J.|last1=Meschi|first2=Rollie J.|last2=Myers|date=30 July 1956|volume=78|issue=24|page=6220|doi=10.1021/ja01605a002}}</ref> |
||
===Preparation=== |
===Preparation=== |
||
Oxidizing [[sulfur]] with [[copper(II) oxide]]:<ref name="Satyanarayana64">{{cite journal|last1=Satyanarayana|first1=S. R.|last2=Vasudeva Murthy|first2=A. R.|year=1964|title=Reactions with Disulphur monoxide Solutions Obtained by the Reduction of Cupric Oxide by Elemental Sulphur|url=http://eprints.iisc.ernet.in/28079/1/32.pdf|journal=Proceedings of the Indian Academy of Sciences, Section A|volume=59|issue=4|pages=263–267|doi=10.1007/BF03046440|s2cid=91428580|access-date=2013-11-08|archive-date=2014-10-17|archive-url=https://web.archive.org/web/20141017050607/http://eprints.iisc.ernet.in/28079/1/32.pdf|url-status=dead}}</ref><ref>{{Cite journal|last1=Dodson|first1=R. M.|last2=Srinivasan|first2=V.|last3=Sharma|first3=K. S.|last4=Sauers|first4=Richard F.|date=July 1972|title=Disulfur monoxide. Reaction with dienes|url=https://pubs.acs.org/doi/pdf/10.1021/jo00980a001|journal=The Journal of Organic Chemistry|volume=37|issue=15|pages=2367–2372|doi=10.1021/jo00980a001|issn=0022-3263}}</ref> |
|||
The reaction of [[thionyl chloride]] with [[silver(I) sulfide]]: |
|||
| ⚫ | |||
A relatively pure generator is the reaction of [[thionyl chloride]] with [[silver(I) sulfide]]:<ref>{{Cite journal|last1=Schenk|first1=P. W.|last2=Steudel|first2=R.|date=1964|title=Preparation of Pure Disulfur Monoxide|url=https://onlinelibrary.wiley.com/doi/abs/10.1002/anie.196400611|journal=Angewandte Chemie International Edition in English|language=en|volume=3|issue=1|pages=61|doi=10.1002/anie.196400611|issn=1521-3773}}</ref> |
|||
:SOCl<sub>2</sub> + Ag<sub>2</sub>S → 2 AgCl + S<sub>2</sub>O |
:SOCl<sub>2</sub> + Ag<sub>2</sub>S → 2 AgCl + S<sub>2</sub>O |
||
Also 5,6-di-''tert''-butyl-2,3,7-trithiabicyclo[2.2.1]hept-5-ene 2-''endo''-7-''endo''-dioxide |
Also 5,6-di-''tert''-butyl-2,3,7-trithiabicyclo[2.2.1]hept-5-ene 2-''endo''-7-''endo''-dioxide decomposes upon heating with release of S<sub>2</sub>O:<ref>{{cite journal|title=Reversible disulfur monoxide (S<sub>2</sub>O)-forming retro-Diels–Alder reaction. disproportionation of S<sub>2</sub>O to trithio-ozone (S<sub>3</sub>) and sulfur dioxide (SO<sub>2</sub>) and reactivities of S<sub>2</sub>O and S<sub>3</sub>|first6=A.|last6=Ishii|first5=Y.|last5=Sugihara|first4=A.|last4=Sakamoto|first3=J.|last3=Takayama|first2=S.|last2=Aoki|last1=Nakayama|first1=J.|journal=Journal of the American Chemical Society|date=28 July 2004|volume=126|issue=29|pmid=15264842|doi=10.1021/ja047729i|pages=9085–9093}}</ref> [[File:S2Ogeneration.svg|center|frameless|upright=2]] |
||
[[File:S2Ogeneration.svg]] |
|||
[[Triphenylphosphine sulfide]] reacts with sulfinyltosylimide to give S<sub>2</sub>O and tosyltriphenylphosphinylamide:<ref>{{cite book|title=Sulfur in Organic and Inorganic Chemistry|volume=1|editor-first=Alexander|editor-last=Senning|year=1971|publisher=Marcel Dekker|location=New York|lccn=70-154612|isbn=0-8247-1615-9|first=Lucreţia|last=Almasi|chapter=The Sulfur–Phosphorus Bond|page=83}}</ref> |
|||
It also arises via thermal decomposition of [[sulfur dioxide]] in a [[glow discharge]].<ref name=candw/> |
|||
:TsNSO + SPPh<sub>3</sub> → TsNPPh<sub>3</sub> + S<sub>2</sub>O |
|||
==Occurrence== |
==Occurrence== |
||
===Volcanism=== |
===Volcanism=== |
||
Volcanoes on [[Io (moon)|Io]] produce substantial quantities of {{Chem|S|2|O}}. It can form between 1% and 6% when hot 100-bar S<sub>2</sub> and SO<sub>2</sub> gas erupts from volcanoes. It is believed that [[Pele (volcano)|Pele]] on Io is surrounded by solid S<sub>2</sub>O.<ref>{{cite journal|url=http://zolotov.faculty.asu.edu/publ/Io-S2O-1998.pdf|first1=Mikhail Yu.|last1=Zolotov|first2=Bruce|last2=Fegley|title=Volcanic Origin of Disulfur Monoxide (S<sub>2</sub>O) on Io|journal=Icarus|volume=133|page=293|date=9 March 1998|doi=10.1006/icar.1998.5930|issue=2|bibcode=1998Icar..133..293Z|access-date=2 October 2010|archive-date=10 October 2017|archive-url=https://web.archive.org/web/20171010200253/http://zolotov.faculty.asu.edu/publ/Io-S2O-1998.pdf|url-status=dead}}</ref> |
|||
===Combustion=== |
|||
===Terran atmosphere=== |
|||
It can be formed by many methods, including combustion of sulfur vapour in a deficiency of oxygen. It arises by oxidizing [[sulfur]] with [[copper(II) oxide]]:<ref name="Satyanarayana64">{{cite journal|last=Satyanarayana|first=S. R.|first2=A. R.|last2=Vasudeva Murthy|year=1964|title=Reactions with Disulphur monoxide Solutions Obtained by the Reduction of Cupric Oxide by Elemental Sulphur|journal=Proceedings of the Indian Academy of Sciences, Section A|volume=59|issue=4|url=http://eprints.iisc.ernet.in/28079/1/32.pdf}}</ref> |
|||
Disulfur monoxide is too unstable to survive at standard conditions,<ref name="Steudel" /> but transient sources include [[incomplete combustion]] of sulfur vapor<ref>{{Cite thesis|last=Khan|first=Ashikur R.|title=Experimental Studies of the Homogenous Conversion of Sulfur Di-Oxide to Sulfur Tri-Oxide via Natural Gas Reburning|date=August 1999|publisher=Ohio University|url=https://etd.ohiolink.edu/apexprod/rws_etd/send_file/send?accession=ohiou1175798586|page=8|quote=Other sulfur oxides are sulfur monoxide, {{chem|S|O}}, its dimer, {{chem|(SO)|z}}, and disulfur monoxide, {{chem|S|2|O}}. These are too unstable or reactive to appear as products of combustion in the ordinary sense, but they are known to occur as intermediates in appropriate circumstances.}}</ref> and thermal decomposition of [[sulfur dioxide]] in a [[glow discharge]].<ref name="candw" /> |
|||
| ⚫ | |||
===As a ligand === |
===As a ligand === |
||
Disulfur monoxide occurs as a [[ligand]] bound to [[transition metal]]s, typically with [[hapticity]] 2.<ref name=":0">{{Cite journal|last1=Urove|first1=Greg A.|last2=Welker|first2=Mark E.|date=April 1988|title=Synthesis of a stable disulfur monoxide precursor and trapping of disulfur monoxide with transition-metal complexes|url=https://pubs.acs.org/doi/pdf/10.1021/om00094a037|journal=Organometallics|volume=7|issue=4|pages=1013–1014|doi=10.1021/om00094a037|issn=0276-7333}}</ref> Examples include {{Chem2|OsCl(NO)(PPh3)2(S2O)}};<ref>{{Cite book|last=Pandey|first=Krishna K.|chapter-url=https://books.google.com/books?id=-mtAYfs9B3YC&pg=PA492|title=Progress in Inorganic Chemistry|date=2009-09-17|publisher=John Wiley & Sons|isbn=978-0-470-16698-7|editor-last=Lippard|editor-first=Stephen J.|volume=80|pages=492|language=en|chapter=Coordination chemistry of thionitrosyl ({{chem|N|S}}), thiazate ({{chem|N|S|O|-}}), disulfidothionitrate ({{chem|S|3|N|-}}), sulfur monoxide ({{chem|S|O}}), and disulfur monoxide ({{chem|S|2|O}}) ligands}}</ref> {{Chem2|[Ir(PPh2)2(S2O)]+}}; and {{Chem2|MeCpMn(CO2)(S2O)}}.<ref name=":0" /> These complexes are closely related to [[transition metal sulfur dioxide complex]]es. |
|||
Disulfur monoxide occurs as a [[ligand]] bound to [[transition metal]]s. One example is OsCl(NO)(PPh<sub>3</sub>)<sub>2</sub>(η<sup>2</sup>-S<sub>2</sub>O). These complexes are closely related to [[transition metal sulfur dioxide complex]]es. |
|||
==Reactions== |
==Reactions== |
||
| ⚫ | |||
S<sub>2</sub>O disproportionates to [[trisulfur]] and [[sulfur dioxide]]: |
|||
:2 S<sub>2</sub>O → S<sub>3</sub> + SO<sub>2</sub> |
:2 S<sub>2</sub>O → "S<sub>3</sub>" + SO<sub>2</sub> |
||
{{Chem|S|2|O}} reacts with [[diazoalkane]]s to form [[dithiirane 1-oxides]].<ref>{{cite journal|last1=Ishii|first1=A.|last2=Kawai|first2=T.|last3=Tekura|first3=K.|last4=Oshida|first4=H.|last5=Nakayama|first5=J.|date=18 May 2001|title=A Convenient Method for the Generation of a Disulfur Monoxide Equivalent and Its Reaction with Diazoalkanes to Yield Dithiirane 1-Oxides|journal=Angewandte Chemie International Edition|volume=40|issue=10|pages=1924–1926|doi=10.1002/1521-3773(20010518)40:10<1924::AID-ANIE1924>3.0.CO;2-F|pmid=11385674}}</ref> |
|||
==Further reading== |
==Further reading== |
||
*Possible biological occurrence: {{cite journal|last1=Iverson|first1=W. P.|title=Disulfur monoxide: production by ''Desulfovibrio''|journal=Science|date=26 May 1967|volume=156|pages=1112–1114|issue=3778|pmid= 6024190|doi=10.1126/science.156.3778.1112|bibcode = 1967Sci...156.1112I |url=http://www.dtic.mil/get-tr-doc/pdf?AD=AD0813804}} |
*Possible biological occurrence: {{cite journal|last1=Iverson|first1=W. P.|title=Disulfur monoxide: production by ''Desulfovibrio''|journal=Science|date=26 May 1967|volume=156|pages=1112–1114|issue=3778|pmid= 6024190|doi=10.1126/science.156.3778.1112|bibcode = 1967Sci...156.1112I |s2cid=3058359|url=http://www.dtic.mil/get-tr-doc/pdf?AD=AD0813804|archive-url=https://web.archive.org/web/20170926033814/http://www.dtic.mil/get-tr-doc/pdf?AD=AD0813804|url-status=dead|archive-date=September 26, 2017}} |
||
*Cyclic disulfur monoxide: {{cite journal|last1=Lo|first1=Wen-Jui|last2=Wu|first2=Yu-Jong|author-link3=Yuan-Pern Lee|last3=Lee|first3=Yuan-Pern|title=Ultraviolet Absorption Spectrum of Cyclic S<sub>2</sub>O in Solid Ar|journal=The Journal of Physical Chemistry A|date=September 2003|volume=107|issue=36|pages=6944–6947|doi=10.1021/jp034563j|bibcode=2003JPCA..107.6944L}} |
*Cyclic disulfur monoxide: {{cite journal|last1=Lo|first1=Wen-Jui|last2=Wu|first2=Yu-Jong|author-link3=Yuan-Pern Lee|last3=Lee|first3=Yuan-Pern|title=Ultraviolet Absorption Spectrum of Cyclic S<sub>2</sub>O in Solid Ar|journal=The Journal of Physical Chemistry A|date=September 2003|volume=107|issue=36|pages=6944–6947|doi=10.1021/jp034563j|bibcode=2003JPCA..107.6944L}} |
||
*Discovery of S<sub>2</sub>O: {{cite journal|last1=Schenk|first1=Peter W.|title=Über das Schwefelmonoxyd|trans-title=On sulfur monoxide|journal=Zeitschrift für Anorganische und Allgemeine Chemie|date=18 March 1933|volume=211|issue=1–2|pages=150–160|doi=10.1002/zaac.19332110117|language=de}} |
*Discovery of S<sub>2</sub>O: {{cite journal|last1=Schenk|first1=Peter W.|title=Über das Schwefelmonoxyd|trans-title=On sulfur monoxide|journal=Zeitschrift für Anorganische und Allgemeine Chemie|date=18 March 1933|volume=211|issue=1–2|pages=150–160|doi=10.1002/zaac.19332110117|language=de}} |
||
Latest revision as of 15:01, 15 November 2024
| |
| |
| Names | |
|---|---|
| Other names
sulfur suboxide; sulfuroxide;
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| Properties | |
| S2O | |
| Molar mass | 80.1294 g/mol[1] |
| Appearance | colourless gas or dark red solid[2] |
| Structure | |
| bent | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
toxic |
| Related compounds | |
Related compounds
|
Trisulfur SO Ozone SO2 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
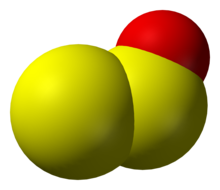
Disulfur monoxide or sulfur suboxide is an inorganic compound with the formula S2O, one of the lower sulfur oxides. It is a colourless gas and condenses to give a roughly dark red coloured solid that is unstable at room temperature.
S
2O occurs rarely in natural atmospheres, but can be made by a variety of laboratory procedures. For this reason, its spectroscopic signature is very well understood.
Structure and spectrum
[edit]Like sulfur dioxide (and, indeed, most molecules) but unlike sulfur monoxide, disulfur, or dioxygen, the ground state of disulfur monoxide is a singlet.[3][4]
Condensed solid S2O absorbs at 420 nm (roughly indigo) and 530 nm (roughly lime). These bands have been assigned to decomposition products S3 and S4.[5]
In the ultraviolet, S2O has absorption band systems in the ranges 250–340 nm and 190–240 nm. There are bands at 323.5 and 327.8 nm.[6] The band in the 315–340 nm range is due to the C1A′–X1A′ (π* ← π) transition.[7]
Gaseous disulfur monoxide does not absorb light in the visible spectrum.
The microwave spectrum of S2O has the following rotational parameters: A = 41915.44 MHz, B = 5059.07 MHz, and C = 4507.19 MHz.[8] Moreover, the microwave spectrum suggests the S−S−O angle is 117.88° with S−S and S−O bond lengths of 188.4 and 146.5 pm, respectively.[9] In the 327.8 nm excited state, the central angle tightens to 109°.[6]
The harmonic frequency for S−S stretching is 415.2 cm−1.[7]
Synthesis
[edit]Historical
[edit]Disulfur monoxide was discovered by Peter W. Schenk in 1933[10] with a glow discharge though sulfur vapour and sulfur dioxide. He discovered that the gas could survive for hours at single digit pressures of mercury in clean glass, but it decomposed near 30 mmHg (4 kPa). Schenk assigned the formula as SO and called it sulfur monoxide. In 1956, D. J. Meschi and R. J. Myers established the formula as S2O.[11]
Preparation
[edit]Oxidizing sulfur with copper(II) oxide:[12][13]
- 3 S8 + 12 CuO → 12 CuS + 4 S2O + 4 SO2
A relatively pure generator is the reaction of thionyl chloride with silver(I) sulfide:[14]
- SOCl2 + Ag2S → 2 AgCl + S2O
Also 5,6-di-tert-butyl-2,3,7-trithiabicyclo[2.2.1]hept-5-ene 2-endo-7-endo-dioxide decomposes upon heating with release of S2O:[15]
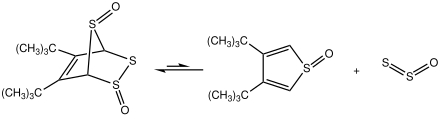
Triphenylphosphine sulfide reacts with sulfinyltosylimide to give S2O and tosyltriphenylphosphinylamide:[16]
- TsNSO + SPPh3 → TsNPPh3 + S2O
Occurrence
[edit]Volcanism
[edit]Volcanoes on Io produce substantial quantities of S
2O. It can form between 1% and 6% when hot 100-bar S2 and SO2 gas erupts from volcanoes. It is believed that Pele on Io is surrounded by solid S2O.[17]
Terran atmosphere
[edit]Disulfur monoxide is too unstable to survive at standard conditions,[10] but transient sources include incomplete combustion of sulfur vapor[18] and thermal decomposition of sulfur dioxide in a glow discharge.[19]
As a ligand
[edit]Disulfur monoxide occurs as a ligand bound to transition metals, typically with hapticity 2.[20] Examples include OsCl(NO)(PPh3)2(S2O);[21] [Ir(PPh2)2(S2O)]+; and MeCpMn(CO2)(S2O).[20] These complexes are closely related to transition metal sulfur dioxide complexes.
Reactions
[edit]On decomposition at room temperature it forms SO2 via the formation of polysulfur oxides:[19]
- 2 S2O → "S3" + SO2
S
2O reacts with diazoalkanes to form dithiirane 1-oxides.[22]
Further reading
[edit]- Possible biological occurrence: Iverson, W. P. (26 May 1967). "Disulfur monoxide: production by Desulfovibrio". Science. 156 (3778): 1112–1114. Bibcode:1967Sci...156.1112I. doi:10.1126/science.156.3778.1112. PMID 6024190. S2CID 3058359. Archived from the original on September 26, 2017.
- Cyclic disulfur monoxide: Lo, Wen-Jui; Wu, Yu-Jong; Lee, Yuan-Pern (September 2003). "Ultraviolet Absorption Spectrum of Cyclic S2O in Solid Ar". The Journal of Physical Chemistry A. 107 (36): 6944–6947. Bibcode:2003JPCA..107.6944L. doi:10.1021/jp034563j.
- Discovery of S2O: Schenk, Peter W. (18 March 1933). "Über das Schwefelmonoxyd" [On sulfur monoxide]. Zeitschrift für Anorganische und Allgemeine Chemie (in German). 211 (1–2): 150–160. doi:10.1002/zaac.19332110117.
References
[edit]- ^ a b c "Disulfur monoxide". NIST. 2008.
- ^ Hapke, B.; Graham, F. (May 1989). "Spectral properties of condensed phases of disulfur monoxide, polysulfur oxide, and irradiated sulfur". Icarus. 79 (1): 47. Bibcode:1989Icar...79...47H. doi:10.1016/0019-1035(89)90107-3.
- ^ Clouthier, Dennis J.; Rutherford, Michael L. (1988) [13 May 1988], "Supersonic jet spectroscopy of S2O", Chemical Physics, 127 (1–3), Amsterdam: North-Holland: 190, Bibcode:1988CP....127..189C, doi:10.1016/0301-0104(88)87117-9
- ^ Han Huixian; Suo Bingbing; Jiang Zhenyi; Wang Yubin; Wen Zhenyi (2008) [7 Jan 2008]. "The potential energy curves of low-lying electronic states of S2O". The Journal of Chemical Physics. 128 (18). American Institute of Physics. Fig. 2. Bibcode:2008JChPh.128r4312H. doi:10.1063/1.2917236. PMID 18532817.
- ^ a b Hallin, K-E. J.; Merer, A. J.; Milton, D. J. (November 1977). "Rotational analysis of bands of the 3400 Å system of disulphur monoxide (S2O)". Canadian Journal of Physics. 55 (21): 1858–1867. Bibcode:1977CaJPh..55.1858H. doi:10.1139/p77-226.
- ^ a b Zhang, Qingguo; Dupré, Patrick; Grzybowski, Bartosz; Vaccaro, Patrick H. (1995). "Laser-induced fluorescence studies of jet-cooled S2O: Axis-switching and predissociation effects". The Journal of Chemical Physics. 103 (1): 67. Bibcode:1995JChPh.103...67Z. doi:10.1063/1.469623.
- ^ Cook, Robert L; Winnewisser, Gisbert; Lindsey, D.C (May 1973). "The centrifugal distortion constants of disulfur monoxide". Journal of Molecular Spectroscopy. 46 (2): 276–284. Bibcode:1973JMoSp..46..276C. doi:10.1016/0022-2852(73)90042-8.
- ^ Meschi, D. J.; Myers, R. J. (1959). "The microwave spectrum, structure, and dipole moment of disulfur monoxide". Journal of Molecular Spectroscopy. 3 (1–6): 405–416. Bibcode:1959JMoSp...3..405M. doi:10.1016/0022-2852(59)90036-0.
- ^ a b Steudel, R. (2003). "Sulfur-Rich Oxides SnO and SnO2". In Steudel, R. (ed.). Elemental Sulfur and Sulfur-Rich Compounds II. Berlin/Heidelberg: Springer. ISBN 9783540449515.
- ^ Meschi, David J.; Myers, Rollie J. (30 July 1956). "Disulfur Monoxide. I. Its Identification as the Major Constituent in Schenk's "Sulfur Monoxide"". Journal of the American Chemical Society. 78 (24): 6220. doi:10.1021/ja01605a002.
- ^ Satyanarayana, S. R.; Vasudeva Murthy, A. R. (1964). "Reactions with Disulphur monoxide Solutions Obtained by the Reduction of Cupric Oxide by Elemental Sulphur" (PDF). Proceedings of the Indian Academy of Sciences, Section A. 59 (4): 263–267. doi:10.1007/BF03046440. S2CID 91428580. Archived from the original (PDF) on 2014-10-17. Retrieved 2013-11-08.
- ^ Dodson, R. M.; Srinivasan, V.; Sharma, K. S.; Sauers, Richard F. (July 1972). "Disulfur monoxide. Reaction with dienes". The Journal of Organic Chemistry. 37 (15): 2367–2372. doi:10.1021/jo00980a001. ISSN 0022-3263.
- ^ Schenk, P. W.; Steudel, R. (1964). "Preparation of Pure Disulfur Monoxide". Angewandte Chemie International Edition in English. 3 (1): 61. doi:10.1002/anie.196400611. ISSN 1521-3773.
- ^ Nakayama, J.; Aoki, S.; Takayama, J.; Sakamoto, A.; Sugihara, Y.; Ishii, A. (28 July 2004). "Reversible disulfur monoxide (S2O)-forming retro-Diels–Alder reaction. disproportionation of S2O to trithio-ozone (S3) and sulfur dioxide (SO2) and reactivities of S2O and S3". Journal of the American Chemical Society. 126 (29): 9085–9093. doi:10.1021/ja047729i. PMID 15264842.
- ^ Almasi, Lucreţia (1971). "The Sulfur–Phosphorus Bond". In Senning, Alexander (ed.). Sulfur in Organic and Inorganic Chemistry. Vol. 1. New York: Marcel Dekker. p. 83. ISBN 0-8247-1615-9. LCCN 70-154612.
- ^ Zolotov, Mikhail Yu.; Fegley, Bruce (9 March 1998). "Volcanic Origin of Disulfur Monoxide (S2O) on Io" (PDF). Icarus. 133 (2): 293. Bibcode:1998Icar..133..293Z. doi:10.1006/icar.1998.5930. Archived from the original (PDF) on 10 October 2017. Retrieved 2 October 2010.
- ^ Khan, Ashikur R. (August 1999). Experimental Studies of the Homogenous Conversion of Sulfur Di-Oxide to Sulfur Tri-Oxide via Natural Gas Reburning (Thesis). Ohio University. p. 8.
Other sulfur oxides are sulfur monoxide, SO, its dimer, (SO)
z, and disulfur monoxide, S
2O. These are too unstable or reactive to appear as products of combustion in the ordinary sense, but they are known to occur as intermediates in appropriate circumstances. - ^ a b Cotton and Wilkinson (1966). Advanced Inorganic Chemistry: A Comprehensive Treatise. p. 540.
- ^ a b Urove, Greg A.; Welker, Mark E. (April 1988). "Synthesis of a stable disulfur monoxide precursor and trapping of disulfur monoxide with transition-metal complexes". Organometallics. 7 (4): 1013–1014. doi:10.1021/om00094a037. ISSN 0276-7333.
- ^ Pandey, Krishna K. (2009-09-17). "Coordination chemistry of thionitrosyl (NS), thiazate (NSO−
), disulfidothionitrate (S
3N−
), sulfur monoxide (SO), and disulfur monoxide (S
2O) ligands". In Lippard, Stephen J. (ed.). Progress in Inorganic Chemistry. Vol. 80. John Wiley & Sons. p. 492. ISBN 978-0-470-16698-7. - ^ Ishii, A.; Kawai, T.; Tekura, K.; Oshida, H.; Nakayama, J. (18 May 2001). "A Convenient Method for the Generation of a Disulfur Monoxide Equivalent and Its Reaction with Diazoalkanes to Yield Dithiirane 1-Oxides". Angewandte Chemie International Edition. 40 (10): 1924–1926. doi:10.1002/1521-3773(20010518)40:10<1924::AID-ANIE1924>3.0.CO;2-F. PMID 11385674.
