Herpes: Difference between revisions
Rreagan007 (talk | contribs) No edit summary |
|||
| (17 intermediate revisions by 12 users not shown) | |||
| Line 3: | Line 3: | ||
{{For|all types of herpes viruses|Herpesviridae}}{{other uses}} |
{{For|all types of herpes viruses|Herpesviridae}}{{other uses}} |
||
}} |
}} |
||
| ⚫ | |||
{{Pp-semi-indef}} |
{{Pp-semi-indef}} |
||
{{Pp-move|small=yes}} |
{{Pp-move|small=yes}} |
||
| ⚫ | |||
{{Infobox medical condition (new) |
{{Infobox medical condition (new) |
||
| synonyms = Herpes simplex |
| synonyms = Herpes simplex |
||
| Line 30: | Line 30: | ||
}} |
}} |
||
'''Herpes simplex''', often known simply as '''herpes''', is a [[viral disease|viral infection]] caused by the [[herpes simplex virus]].<ref name="WHO2022">{{cite web |title=Herpes simplex virus |url=https://www.who.int/news-room/fact-sheets/detail/herpes-simplex-virus |website=World Health Organization |access-date=21 November 2022}}</ref> Herpes infections are categorized by the area of the body that is infected. The two major types of herpes are [[Cold sore|oral herpes]] and [[genital herpes]], though [[ |
'''Herpes simplex''', often known simply as '''herpes''', is a [[viral disease|viral infection]] caused by the [[herpes simplex virus]].<ref name="WHO2022">{{cite web |title=Herpes simplex virus |url=https://www.who.int/news-room/fact-sheets/detail/herpes-simplex-virus |website=World Health Organization |access-date=21 November 2022}}</ref> Herpes infections are categorized by the area of the body that is infected. The two major types of herpes are [[Cold sore|oral herpes]] and [[genital herpes]], though [[Herpes simplex#Types of herpes|other forms]] also exist. |
||
Oral herpes involves the face or mouth. It may result in small [[blister]]s in groups, often called cold sores or fever blisters, or may just cause a sore throat.<ref name=Bal2014/><ref>{{cite book|last1=Mosby|title=Mosby's Medical Dictionary|date=2013|publisher=Elsevier Health Sciences|isbn=9780323112581|pages=836–37|edition=9|url=https://books.google.com/books?id=aW0zkZl0JgQC&pg=PA836|url-status=live|archive-url=https://web.archive.org/web/20170906210724/https://books.google.com/books?id=aW0zkZl0JgQC&pg=PA836|archive-date=2017-09-06}}</ref> Genital herpes involves the [[genitalia]]. It may have minimal symptoms or form blisters that break open and result in small [[ulcer]]s.<ref name=CDC2014F/> These typically heal over two to four weeks.<ref name=CDC2014F/> Tingling or shooting pains may occur before the blisters appear.<ref name=CDC2014F/> |
Oral herpes involves the face or mouth. It may result in small [[blister]]s in groups, often called cold sores or fever blisters, or may just cause a sore throat.<ref name=Bal2014/><ref>{{cite book|last1=Mosby|title=Mosby's Medical Dictionary|date=2013|publisher=Elsevier Health Sciences|isbn=9780323112581|pages=836–37|edition=9|url=https://books.google.com/books?id=aW0zkZl0JgQC&pg=PA836|url-status=live|archive-url=https://web.archive.org/web/20170906210724/https://books.google.com/books?id=aW0zkZl0JgQC&pg=PA836|archive-date=2017-09-06}}</ref> Genital herpes involves the [[genitalia]]. It may have minimal symptoms or form blisters that break open and result in small [[ulcer]]s.<ref name=CDC2014F/> These typically heal over two to four weeks.<ref name=CDC2014F/> Tingling or shooting pains may occur before the blisters appear.<ref name=CDC2014F/> |
||
Herpes cycles between periods of active disease followed by periods without symptoms.<ref name=CDC2014F/> The first episode is often more severe and may be associated with fever, muscle pains, swollen [[lymph node]]s and headaches.<ref name=CDC2014F/> Over time, episodes of active disease decrease in frequency and severity.<ref name=CDC2014F/> |
Herpes cycles between periods of active disease followed by periods without symptoms.<ref name=CDC2014F/> The first episode is often more severe and may be associated with fever, muscle pains, swollen [[lymph node]]s and headaches.<ref name=CDC2014F/> Over time, episodes of active disease decrease in frequency and severity.<ref name=CDC2014F/> |
||
| Line 42: | Line 42: | ||
The most effective method of avoiding genital infections is by avoiding vaginal, oral, manual, and anal sex.<ref name=CDC2014F/><ref>{{cite book| last1 = Hoyle | first1 = Alice | last2 = McGeeney | first2 = Ester |title=Great Relationships and Sex Education|publisher=Taylor and Francis|year=2019|access-date=July 11, 2023|isbn=978-1-35118-825-8|url=https://books.google.com/books?id=KE7ADwAAQBAJ&pg=PT261}}</ref> [[Condom]] use decreases the risk.<ref name=CDC2014F/> Daily [[antiviral drug|antiviral medication]] taken by someone who has the infection can also reduce spread.<ref name=CDC2014F/> There is no available [[vaccine]]<ref name=CDC2014F/> and once infected, there is no cure.<ref name=CDC2014F/> [[Paracetamol]] (acetaminophen) and topical lidocaine may be used to help with the symptoms.<ref name=Bal2014/> Treatments with antiviral medication such as [[aciclovir]] or [[valaciclovir]] can lessen the severity of symptomatic episodes.<ref name=CDC2014F/><ref name=Bal2014/> |
The most effective method of avoiding genital infections is by avoiding vaginal, oral, manual, and anal sex.<ref name=CDC2014F/><ref>{{cite book| last1 = Hoyle | first1 = Alice | last2 = McGeeney | first2 = Ester |title=Great Relationships and Sex Education|publisher=Taylor and Francis|year=2019|access-date=July 11, 2023|isbn=978-1-35118-825-8|url=https://books.google.com/books?id=KE7ADwAAQBAJ&pg=PT261}}</ref> [[Condom]] use decreases the risk.<ref name=CDC2014F/> Daily [[antiviral drug|antiviral medication]] taken by someone who has the infection can also reduce spread.<ref name=CDC2014F/> There is no available [[vaccine]]<ref name=CDC2014F/> and once infected, there is no cure.<ref name=CDC2014F/> [[Paracetamol]] (acetaminophen) and topical lidocaine may be used to help with the symptoms.<ref name=Bal2014/> Treatments with antiviral medication such as [[aciclovir]] or [[valaciclovir]] can lessen the severity of symptomatic episodes.<ref name=CDC2014F/><ref name=Bal2014/> |
||
Worldwide rates of either HSV-1 or HSV-2 are between 60% and 95% in adults.<ref name=Peds09/> HSV-1 is usually acquired during childhood.<ref name=CDC2014F>{{cite web |title=Genital Herpes – CDC Fact Sheet |url=https://www.cdc.gov/std/herpes/STDFact-Herpes-detailed.htm |website=cdc.gov |access-date=31 December 2014 |date=December 8, 2014 |url-status=live |archive-url=https://web.archive.org/web/20141231122747/http://www.cdc.gov/std/herpes/STDFact-Herpes-detailed.htm |archive-date=31 December 2014}}</ref> Since there is no cure for either HSV-1 or HSV-2, rates of both inherently increase as people age.<ref name=Peds09/> Rates of HSV-1 are between 70% and 80% in populations of low socioeconomic status and 40% to 60% in populations of improved socioeconomic status.<ref name=Peds09>{{cite journal |vauthors=Chayavichitsilp P, Buckwalter JV, Krakowski AC, Friedlander SF |title=Herpes simplex |journal=Pediatr Rev |volume=30 |issue=4 |pages=119–29; quiz 130 |date=April 2009 |pmid=19339385 |doi=10.1542/pir.30-4-119 |s2cid=34735917 |
Worldwide rates of either HSV-1 or HSV-2 are between 60% and 95% in adults.<ref name=Peds09/> HSV-1 is usually acquired during childhood.<ref name=CDC2014F>{{cite web |title=Genital Herpes – CDC Fact Sheet |url=https://www.cdc.gov/std/herpes/STDFact-Herpes-detailed.htm |website=cdc.gov |access-date=31 December 2014 |date=December 8, 2014 |url-status=live |archive-url=https://web.archive.org/web/20141231122747/http://www.cdc.gov/std/herpes/STDFact-Herpes-detailed.htm |archive-date=31 December 2014}}</ref> Since there is no cure for either HSV-1 or HSV-2, rates of both inherently increase as people age.<ref name=Peds09/> Rates of HSV-1 are between 70% and 80% in populations of low socioeconomic status and 40% to 60% in populations of improved socioeconomic status.<ref name=Peds09>{{cite journal |vauthors=Chayavichitsilp P, Buckwalter JV, Krakowski AC, Friedlander SF |title=Herpes simplex |journal=Pediatr Rev |volume=30 |issue=4 |pages=119–29; quiz 130 |date=April 2009 |pmid=19339385 |doi=10.1542/pir.30-4-119 |s2cid=34735917 }}</ref> An estimated 536 million people worldwide (16% of the population) were infected with HSV-2 as of 2003 with greater rates among women and those in the developing world.<ref name=Looker2008>{{cite journal|last=Looker|first=KJ|author2=Garnett, GP |author3=Schmid, GP |title=An estimate of the global prevalence and incidence of herpes simplex virus type 2 infection.|journal=Bulletin of the World Health Organization|date=October 2008|volume=86|issue=10|pages=805–12, A|pmid=18949218|pmc=2649511|doi=10.2471/blt.07.046128}}</ref> Most people with HSV-2 do not realize that they are infected.<ref name=CDC2014F/> |
||
{{TOC limit|3}} |
{{TOC limit|3}} |
||
==Etymology== |
==Etymology== |
||
The name is from {{ |
The name is from {{langx|grc|ἕρπης}} ''herpēs'', which is related to the meaning 'to creep', referring to spreading blisters.<ref>{{cite journal |last1=Beswick |first1=TSL |title=The Origin and the Use of the Word Herpes |journal=Med Hist |volume=6 |date=1962 |issue=3 |pages=214–232|doi=10.1017/S002572730002737X |pmid=13868599 |pmc=1034725 }}</ref> The name does not refer to latency.<ref>{{cite web |last1=Reese |first1=Vail |title=Countering Creeping Confusion: A Proposal to Re-Name Herpes Virus TAXONOMY |url=http://ojcpcd.com/reese-v/countering-creeping-confusion-a-proposal-to-re-name-herpes-virus-taxonomy/ |website=Online Journal of Community and Person-Centered Dermatology |publisher=Dr. David Elpern |access-date=22 September 2018 |archive-date=23 September 2018 |archive-url=https://web.archive.org/web/20180923005836/http://ojcpcd.com/reese-v/countering-creeping-confusion-a-proposal-to-re-name-herpes-virus-taxonomy/ |url-status=dead }}</ref> |
||
== Signs and symptoms == |
== Signs and symptoms == |
||
[[File:Herpes Infection.png|thumb|Herpes infection]] |
[[File:Herpes Infection.png|thumb|Herpes infection]] |
||
| Line 92: | Line 92: | ||
|} |
|} |
||
=== Other === |
=== Other === |
||
[[Neonatal herpes simplex]] is an HSV infection in an infant. It is a rare but serious condition, usually caused by [[vertical transmission]] of HSV-1 or -2 from mother to newborn. During immunodeficiency, herpes simplex can cause unusual lesions in the skin. One of the most striking is the appearance of clean linear erosions in skin creases, with the appearance of a knife cut.<ref>{{cite journal|title=Linear erosive Herpes Simplex Virus infection in immunocompromised patients: the "Knife-Cut Sign"|journal=Clin Infect Dis|year=2008|volume=47|pages=1440–41|doi=10.1086/592976|author1=Jocelyn A. Lieb |author2=Stacey Brisman |author3=Sara Herman |author4=Jennifer MacGregor |author5=Marc E. Grossman |pmid=18937574|issue=11|doi-access= |
[[Neonatal herpes simplex]] is an HSV infection in an infant. It is a rare but serious condition, usually caused by [[vertical transmission]] of HSV-1 or -2 from mother to newborn. During immunodeficiency, herpes simplex can cause unusual lesions in the skin. One of the most striking is the appearance of clean linear erosions in skin creases, with the appearance of a knife cut.<ref>{{cite journal|title=Linear erosive Herpes Simplex Virus infection in immunocompromised patients: the "Knife-Cut Sign"|journal=Clin Infect Dis|year=2008|volume=47|pages=1440–41|doi=10.1086/592976|author1=Jocelyn A. Lieb |author2=Stacey Brisman |author3=Sara Herman |author4=Jennifer MacGregor |author5=Marc E. Grossman |pmid=18937574|issue=11|doi-access=}}</ref> [[Herpetic sycosis]] is a recurrent or initial herpes simplex infection affecting primarily the hair follicles.<ref name="Andrews">{{cite book|author=James, William D. |title=Andrews' Diseases of the Skin: clinical Dermatology |publisher=Saunders Elsevier |year=2006 |isbn=978-0-7216-2921-6 |last2=Berger |first2=Timothy G. }}</ref>{{rp|369}} [[Eczema herpeticum]] is an infection with herpesvirus in patients with chronic [[atopic dermatitis]] may result in spread of herpes simplex throughout the eczematous areas.<ref name="Andrews" />{{rp|373}} |
||
[[Herpetic keratoconjunctivitis]], a primary infection, typically presents as swelling of the [[conjunctiva]] and eyelids ([[blepharoconjunctivitis]]), accompanied by small white itchy lesions on the surface of the [[cornea]]. |
[[Herpetic keratoconjunctivitis]], a primary infection, typically presents as swelling of the [[conjunctiva]] and eyelids ([[blepharoconjunctivitis]]), accompanied by small white itchy lesions on the surface of the [[cornea]]. |
||
| Line 104: | Line 104: | ||
Although the exact cause of [[Bell's palsy]]{{mdash}}a type of facial [[paralysis]]{{mdash}}is unknown, it may be related to the reactivation of HSV-1.<ref>{{cite journal |vauthors=Tankéré F, Bernat I |title=[Bell's palsy: from viral aetiology to diagnostic reality] |language=fr |journal=Rev Méd Interne |volume=30 |issue=9 |pages=769–75 |date=September 2009 |pmid=19195745 |doi=10.1016/j.revmed.2008.12.006 }}</ref> This theory has been contested, however, since HSV is detected in large numbers of individuals having never experienced facial paralysis, and higher levels of antibodies for HSV are not found in HSV-infected individuals with Bell's palsy compared to those without.<ref name="pmid15699730">{{cite journal |vauthors=Linder T, Bossart W, Bodmer D |title=Bell's palsy and Herpes simplex virus: fact or mystery? |journal=Otol. Neurotol. |volume=26 |issue=1 |pages=109–13 |date=January 2005 |pmid=15699730 |doi= 10.1097/00129492-200501000-00020|s2cid=33873521 }}</ref> Antivirals may improve the condition slightly when used together with [[corticosteroids]] in those with severe disease.<ref>{{Cite journal|last1=Gagyor|first1=Ildiko|last2=Madhok|first2=Vishnu B.|last3=Daly|first3=Fergus|last4=Somasundara|first4=Dhruvashree|last5=Sullivan|first5=Michael|last6=Gammie|first6=Fiona|last7=Sullivan|first7=Frank|date=2015-11-09|title=Antiviral treatment for Bell's palsy (idiopathic facial paralysis)|url=http://discovery.dundee.ac.uk/ws/files/743606/Lockhart_2010.pdf|journal=The Cochrane Database of Systematic Reviews|issue=11|pages=CD001869|doi=10.1002/14651858.CD001869.pub8|issn=1469-493X|pmid=26559436}}</ref> |
Although the exact cause of [[Bell's palsy]]{{mdash}}a type of facial [[paralysis]]{{mdash}}is unknown, it may be related to the reactivation of HSV-1.<ref>{{cite journal |vauthors=Tankéré F, Bernat I |title=[Bell's palsy: from viral aetiology to diagnostic reality] |language=fr |journal=Rev Méd Interne |volume=30 |issue=9 |pages=769–75 |date=September 2009 |pmid=19195745 |doi=10.1016/j.revmed.2008.12.006 }}</ref> This theory has been contested, however, since HSV is detected in large numbers of individuals having never experienced facial paralysis, and higher levels of antibodies for HSV are not found in HSV-infected individuals with Bell's palsy compared to those without.<ref name="pmid15699730">{{cite journal |vauthors=Linder T, Bossart W, Bodmer D |title=Bell's palsy and Herpes simplex virus: fact or mystery? |journal=Otol. Neurotol. |volume=26 |issue=1 |pages=109–13 |date=January 2005 |pmid=15699730 |doi= 10.1097/00129492-200501000-00020|s2cid=33873521 }}</ref> Antivirals may improve the condition slightly when used together with [[corticosteroids]] in those with severe disease.<ref>{{Cite journal|last1=Gagyor|first1=Ildiko|last2=Madhok|first2=Vishnu B.|last3=Daly|first3=Fergus|last4=Somasundara|first4=Dhruvashree|last5=Sullivan|first5=Michael|last6=Gammie|first6=Fiona|last7=Sullivan|first7=Frank|date=2015-11-09|title=Antiviral treatment for Bell's palsy (idiopathic facial paralysis)|url=http://discovery.dundee.ac.uk/ws/files/743606/Lockhart_2010.pdf|journal=The Cochrane Database of Systematic Reviews|issue=11|pages=CD001869|doi=10.1002/14651858.CD001869.pub8|issn=1469-493X|pmid=26559436}}</ref> |
||
=== Alzheimer's disease === |
=== Alzheimer's disease === |
||
HSV-1 has been proposed as a possible cause of [[Alzheimer's disease]].<ref>{{cite journal |vauthors=Itzhaki RF, Wozniak MA |title=Herpes simplex virus type 1 in Alzheimer's disease: the enemy within |journal=J. Alzheimers Dis. |volume=13 |issue=4 |pages=393–405 |date=May 2008 |pmid=18487848 |doi=10.3233/JAD-2008-13405 }}</ref><ref>{{cite journal |vauthors=Holmes C, Cotterell D |title=Role of infection in the pathogenesis of Alzheimer's disease: implications for treatment |journal=CNS Drugs |volume=23 |issue=12 |pages=993–1002 |date=December 2009 |pmid=19958038 |doi=10.2165/11310910-000000000-00000 |s2cid=25248989 }}</ref> In the presence of a certain gene variation ([[Apolipoprotein E|APOE]]-epsilon4 allele carriers), HSV-1 appears to be particularly damaging to the nervous system and increases one's risk of developing Alzheimer's disease. The virus interacts with the components and receptors of [[lipoproteins]], which may lead to its development.<ref name="Dobson1999">{{cite journal|vauthors=Dobson CB, Itzhaki RF |title=Herpes simplex virus type 1 and Alzheimer's disease |journal=Neurobiol. Aging |volume=20 |issue=4 |pages=457–65 |year=1999 |pmid=10604441 |doi=10.1016/S0197-4580(99)00055-X|s2cid=23633290 }}</ref><ref>{{cite journal|author=Pyles RB |title=The association of herpes simplex virus and Alzheimer's disease: a potential synthesis of genetic and environmental factors |journal=Herpes |volume=8 |issue=3 |pages=64–68 |year=2001 |pmid=11867022}}</ref> |
HSV-1 has been proposed as a possible cause of [[Alzheimer's disease]].<ref>{{cite journal |vauthors=Itzhaki RF, Wozniak MA |title=Herpes simplex virus type 1 in Alzheimer's disease: the enemy within |journal=J. Alzheimers Dis. |volume=13 |issue=4 |pages=393–405 |date=May 2008 |pmid=18487848 |doi=10.3233/JAD-2008-13405 }}</ref><ref>{{cite journal |vauthors=Holmes C, Cotterell D |title=Role of infection in the pathogenesis of Alzheimer's disease: implications for treatment |journal=CNS Drugs |volume=23 |issue=12 |pages=993–1002 |date=December 2009 |pmid=19958038 |doi=10.2165/11310910-000000000-00000 |s2cid=25248989 }}</ref> In the presence of a certain gene variation ([[Apolipoprotein E|APOE]]-epsilon4 allele carriers), HSV-1 appears to be particularly damaging to the nervous system and increases one's risk of developing Alzheimer's disease. The virus interacts with the components and receptors of [[lipoproteins]], which may lead to its development.<ref name="Dobson1999">{{cite journal|vauthors=Dobson CB, Itzhaki RF |title=Herpes simplex virus type 1 and Alzheimer's disease |journal=Neurobiol. Aging |volume=20 |issue=4 |pages=457–65 |year=1999 |pmid=10604441 |doi=10.1016/S0197-4580(99)00055-X|s2cid=23633290 }}</ref><ref>{{cite journal|author=Pyles RB |title=The association of herpes simplex virus and Alzheimer's disease: a potential synthesis of genetic and environmental factors |journal=Herpes |volume=8 |issue=3 |pages=64–68 |year=2001 |pmid=11867022}}</ref> |
||
| Line 153: | Line 153: | ||
==Prevention== |
==Prevention== |
||
| ⚫ | As with almost all sexually transmitted infections, women are more susceptible to acquiring genital HSV-2 than men.<ref>{{cite news |author=Carla K. Johnson |title=Percentage of people with herpes drops |url=http://www.aegis.com/news/ap/2006/ap060840.html |access-date=2011-04-12 |agency=Associated Press |date= August 23, 2006 |archive-url=https://web.archive.org/web/20120318125331/http://www.aegis.com/news/ap/2006/ap060840.html |archive-date=2012-03-18 |url-status=dead}}</ref> On an annual basis, without the use of antivirals or condoms, the transmission risk of HSV-2 from infected male to female is about 8–11%.<ref name=Mertz1993/><ref name=Kulhanjian1992>{{cite journal |doi=10.1056/NEJM199204023261403 |author=Kulhanjian J A |title=Identification of women at unsuspected risk of primary infection with herpes simplex virus type 2 during pregnancy |journal=N. Engl. J. Med. |volume=326 |issue=14 |pages=916–20 |pmid=1311799 |date=April 2, 1992 |name-list-style=vanc |author2=Soroush V |author3=Au DS |display-authors=3 |last4=Bronzan |first4=Rachel N. |last5=Yasukawa |first5=Linda L. |last6=Weylman |first6=Laura E. |last7=Arvin |first7=Ann M. |last8=Prober |first8=Charles G. |doi-access=free}}</ref> This is believed to be due to the increased exposure of mucosal tissue to potential infection sites. Transmission risk from infected female to male is around 4–5% annually.<ref name=Kulhanjian1992/> Suppressive antiviral therapy reduces these risks by 50%.<ref name=Corey2004>{{cite journal|author=Corey L|title=Once-daily valacyclovir to reduce the risk of transmission of genital herpes|journal=N Engl J Med|volume=350|issue=1|pages=11–20|date=January 2004|pmid=14702423|doi=10.1056/NEJMoa035144|name-list-style=vanc|author2=Wald A|author3=Patel R|display-authors=3|last4=Sacks|first4=Stephen L.|last5=Tyring|first5=Stephen K.|last6=Warren|first6=Terri|last7=Douglas|first7=John M.|last8=Paavonen|first8=Jorma|last9=Morrow|first9=R. Ashley|s2cid=21573428|doi-access=free}}</ref> Antivirals also help prevent the development of symptomatic HSV in infection scenarios, meaning the infected partner will be seropositive but symptom-free by about 50%. Condom use also reduces the transmission risk significantly.<ref name=Wald>{{cite journal | vauthors=Wald A, Langenberg AG, Link K, Izu AE, Ashley R, Warren T, Tyring S, ((Douglas JM Jr)), Corey L | title=Effect of condoms on reducing the transmission of herpes simplex virus type 2 from men to women | journal=JAMA | year=2001 | pages=3100–06 | volume=285 | issue=24 | pmid=11427138 | doi=10.1001/jama.285.24.3100 | doi-access= }}</ref><ref name=Wald2005>{{cite journal|author=Wald A|title=The relationship between condom use and herpes simplex virus acquisition|journal=Annals of Internal Medicine|volume=143|issue=10|pages=707–13|date=November 2005|pmid=16287791|name-list-style=vanc|author2=Langenberg AG|author3=Krantz E|display-authors=3|last4=Douglas Jr|first4=JM|last5=Handsfield|first5=HH|last6=Dicarlo|first6=RP|last7=Adimora|first7=AA|last8=Izu|first8=AE|last9=Morrow|first9=RA|doi=10.7326/0003-4819-143-10-200511150-00007|s2cid=37342783}}</ref> Condom use is much more effective at preventing male-to-female transmission than ''vice versa''.<ref name=Wald/> Previous HSV-1 infection may reduce the risk for acquisition of HSV-2 infection among women by a factor of three, although the one study that states this has a small sample size of 14 transmissions out of 214 couples.<ref>{{cite journal |last=Mertz |first=GJ |author2=Benedetti J |author3=Ashley R |author4=Selke SA |author5=Corey L. |title=Risk factors for the sexual transmission of genital herpes |journal=Annals of Internal Medicine |date=1 February 1992 |volume=116 |issue=3 |pages=197–202 |pmid=1309413 |doi=10.7326/0003-4819-116-3-197|s2cid=40143915 }}</ref> |
||
[[File:Kondom.jpg|thumb|Barrier protection, such as a [[condom]], can reduce the risk of herpes transmission.]] |
|||
| ⚫ | As with almost all sexually transmitted infections, women are more susceptible to acquiring genital HSV-2 than men.<ref>{{cite news |author=Carla K. Johnson |title=Percentage of people with herpes drops |url=http://www.aegis.com/news/ap/2006/ap060840.html |access-date=2011-04-12 |agency=Associated Press |date= August 23, 2006 |archive-url=https://web.archive.org/web/20120318125331/http://www.aegis.com/news/ap/2006/ap060840.html |archive-date=2012-03-18 |url-status=dead}}</ref> On an annual basis, without the use of antivirals or condoms, the transmission risk of HSV-2 from infected male to female is about 8–11%.<ref name=Mertz1993/><ref name=Kulhanjian1992>{{cite journal |doi=10.1056/NEJM199204023261403 |author=Kulhanjian J A |title=Identification of women at unsuspected risk of primary infection with herpes simplex virus type 2 during pregnancy |journal=N. Engl. J. Med. |volume=326 |issue=14 |pages=916–20 |pmid=1311799 |date=April 2, 1992 |name-list-style=vanc |author2=Soroush V |author3=Au DS |display-authors=3 |last4=Bronzan |first4=Rachel N. |last5=Yasukawa |first5=Linda L. |last6=Weylman |first6=Laura E. |last7=Arvin |first7=Ann M. |last8=Prober |first8=Charles G. |doi-access=free}}</ref> This is believed to be due to the increased exposure of mucosal tissue to potential infection sites. Transmission risk from infected female to male is around 4–5% annually.<ref name=Kulhanjian1992/> Suppressive antiviral therapy reduces these risks by 50%.<ref name=Corey2004>{{cite journal|author=Corey L|title=Once-daily valacyclovir to reduce the risk of transmission of genital herpes|journal=N Engl J Med|volume=350|issue=1|pages=11–20|date=January 2004|pmid=14702423|doi=10.1056/NEJMoa035144|name-list-style=vanc|author2=Wald A|author3=Patel R|display-authors=3|last4=Sacks|first4=Stephen L.|last5=Tyring|first5=Stephen K.|last6=Warren|first6=Terri|last7=Douglas|first7=John M.|last8=Paavonen|first8=Jorma|last9=Morrow|first9=R. Ashley|s2cid=21573428|doi-access=free}}</ref> Antivirals also help prevent the development of symptomatic HSV in infection scenarios, meaning the infected partner will be seropositive but symptom-free by about 50%. Condom use also reduces the transmission risk significantly.<ref name=Wald>{{cite journal | vauthors=Wald A, Langenberg AG, Link K, Izu AE, Ashley R, Warren T, Tyring S, ((Douglas JM Jr)), Corey L | title=Effect of condoms on reducing the transmission of herpes simplex virus type 2 from men to women | journal=JAMA | year=2001 | pages=3100–06 | volume=285 | issue=24 | pmid=11427138 | doi=10.1001/jama.285.24.3100 | doi-access= |
||
However, asymptomatic carriers of the HSV-2 virus are still contagious. In many infections, the first symptom people will have of their own infections is the horizontal transmission to a sexual partner or the vertical transmission of neonatal herpes to a newborn at term. Since most asymptomatic individuals are unaware of their infection, they are considered at high risk for spreading HSV.<ref>{{cite web |url=https://www.cdc.gov/std/herpes/STDFact-herpes-detailed.htm#ref21 |title=Genital Herpes – CDC Fact Sheet |publisher=Center for Disease Control and Prevention |access-date=2014-01-30 |url-status=live |archive-url=https://web.archive.org/web/20140130165350/http://www.cdc.gov/std/Herpes/STDFact-herpes-detailed.htm#ref21 |archive-date=2014-01-30 }}</ref> |
However, asymptomatic carriers of the HSV-2 virus are still contagious. In many infections, the first symptom people will have of their own infections is the horizontal transmission to a sexual partner or the vertical transmission of neonatal herpes to a newborn at term. Since most asymptomatic individuals are unaware of their infection, they are considered at high risk for spreading HSV.<ref>{{cite web |url=https://www.cdc.gov/std/herpes/STDFact-herpes-detailed.htm#ref21 |title=Genital Herpes – CDC Fact Sheet |publisher=Center for Disease Control and Prevention |access-date=2014-01-30 |url-status=live |archive-url=https://web.archive.org/web/20140130165350/http://www.cdc.gov/std/Herpes/STDFact-herpes-detailed.htm#ref21 |archive-date=2014-01-30 }}</ref> |
||
| Line 161: | Line 160: | ||
===Barrier methods=== |
===Barrier methods=== |
||
Condoms offer moderate protection against HSV-2 in both men and women, with consistent condom users having a 30%-lower risk of HSV-2 acquisition compared with those who never use condoms.<ref name=Martin>{{cite journal | vauthors = Martin ET, Krantz E, Gottlieb SL, Magaret AS, Langenberg A, Stanberry L, Kamb M, Wald A | title = A pooled analysis of the effect of condoms in preventing HSV-2 acquisition | journal = Archives of Internal Medicine | volume = 169 | issue = 13 | pages = 1233–40 | date = July 2009 | pmid = 19597073 | pmc = 2860381 | doi = 10.1001/archinternmed.2009.177 }}</ref> A [[female condom]] can provide greater protection than the male condom, as it covers the labia.<ref> |
Condoms offer moderate protection against HSV-2 in both men and women, with consistent condom users having a 30%-lower risk of HSV-2 acquisition compared with those who never use condoms.<ref name=Martin>{{cite journal | vauthors = Martin ET, Krantz E, Gottlieb SL, Magaret AS, Langenberg A, Stanberry L, Kamb M, Wald A | title = A pooled analysis of the effect of condoms in preventing HSV-2 acquisition | journal = Archives of Internal Medicine | volume = 169 | issue = 13 | pages = 1233–40 | date = July 2009 | pmid = 19597073 | pmc = 2860381 | doi = 10.1001/archinternmed.2009.177 }}</ref> A [[female condom]] can provide greater protection than the male condom, as it covers the labia.<ref>{{cite web | publisher = UBM Medica | url = http://www.obgyn.net/femalepatient/femalepatient.asp?page=herpes_tfp | title = Putting Herpes in Perspective | access-date = 20 July 2011 | archive-date = 25 February 2021 | archive-url = https://web.archive.org/web/20210225150028/https://www.contemporaryobgyn.net/femalepatient/femalepatient.asp?page=herpes_tfp | url-status = dead }}</ref> The virus cannot pass through a synthetic condom, but a male condom's effectiveness is limited<ref>{{cite web|url=https://www.cdc.gov/condomeffectiveness/latex.htm|title=Condom Effectiveness – Male Latex Condoms and Sexually Transmitted Diseases|publisher=Center for Disease Control and Prevention|archive-url=https://web.archive.org/web/20111002073943/http://www.cdc.gov/condomeffectiveness/latex.htm|archive-date=2011-10-02|url-status=live|access-date=2011-10-01}}</ref> because herpes ulcers may appear on areas not covered by it. Neither type of condom prevents contact with the scrotum, anus, buttocks, or upper thighs, areas that may come in contact with ulcers or genital secretions during sexual activity. Protection against herpes simplex depends on the site of the ulcer; therefore, if ulcers appear on areas not covered by condoms, abstaining from sexual activity until the ulcers are fully healed is one way to limit risk of transmission.<ref>{{cite web|url=https://www.cdc.gov/std/herpes/stdfact-herpes.htm|title=STD Facts – Genital Herpes|publisher=Center for Disease Control and Prevention|archive-url=https://web.archive.org/web/20111001182509/http://www.cdc.gov/std/herpes/stdfact-herpes.htm|archive-date=2011-10-01|url-status=live|access-date=2011-10-01}}</ref> The risk is not eliminated, however, as viral shedding capable of transmitting infection may still occur while the infected partner is asymptomatic.<ref>{{cite journal |last=Koelle |first=D.M. |author2=Wald, A. |title=Herpes simplex virus: The importance of asymptomatic shedding |date=April 2000 |journal=J. Antimicrob. Chemother. |volume=45 |issue=Suppl T3 |pages=1–8 |pmid=10855766 |doi=10.1093/jac/45.suppl_4.1|doi-access=free }}</ref> The use of condoms or [[dental dams]] also limits the transmission of herpes from the genitals of one partner to the mouth of the other (or ''vice versa'') during [[oral sex]]. When one partner has a herpes simplex infection and the other does not, the use of antiviral medication, such as [[valaciclovir]], in conjunction with a condom, further decreases the chances of transmission to the uninfected partner.<ref name="pmid18156035"/> Topical [[microbicide]]s that contain chemicals that directly inactivate the virus and block viral entry are being investigated.<ref name="pmid18156035"/> |
||
{{cite web | publisher = UBM Medica | url=http://www.obgyn.net/femalepatient/femalepatient.asp?page=herpes_tfp | title=Putting Herpes in Perspective | access-date=20 July 2011}}</ref> The virus cannot pass through a synthetic condom, but a male condom's effectiveness is limited<ref>{{cite web|url=https://www.cdc.gov/condomeffectiveness/latex.htm|title=Condom Effectiveness – Male Latex Condoms and Sexually Transmitted Diseases|publisher=Center for Disease Control and Prevention|archive-url=https://web.archive.org/web/20111002073943/http://www.cdc.gov/condomeffectiveness/latex.htm|archive-date=2011-10-02|url-status=live|access-date=2011-10-01}}</ref> because herpes ulcers may appear on areas not covered by it. Neither type of condom prevents contact with the scrotum, anus, buttocks, or upper thighs, areas that may come in contact with ulcers or genital secretions during sexual activity. Protection against herpes simplex depends on the site of the ulcer; therefore, if ulcers appear on areas not covered by condoms, abstaining from sexual activity until the ulcers are fully healed is one way to limit risk of transmission.<ref>{{cite web|url=https://www.cdc.gov/std/herpes/stdfact-herpes.htm|title=STD Facts – Genital Herpes|publisher=Center for Disease Control and Prevention|archive-url=https://web.archive.org/web/20111001182509/http://www.cdc.gov/std/herpes/stdfact-herpes.htm|archive-date=2011-10-01|url-status=live|access-date=2011-10-01}}</ref> The risk is not eliminated, however, as viral shedding capable of transmitting infection may still occur while the infected partner is asymptomatic.<ref>{{cite journal |last=Koelle |first=D.M. |author2=Wald, A. |title=Herpes simplex virus: The importance of asymptomatic shedding |date=April 2000 |journal=J. Antimicrob. Chemother. |volume=45 |issue=Suppl T3 |pages=1–8 |pmid=10855766 |doi=10.1093/jac/45.suppl_4.1|doi-access=free }}</ref> The use of condoms or [[dental dams]] also limits the transmission of herpes from the genitals of one partner to the mouth of the other (or ''vice versa'') during [[oral sex]]. When one partner has a herpes simplex infection and the other does not, the use of antiviral medication, such as [[valaciclovir]], in conjunction with a condom, further decreases the chances of transmission to the uninfected partner.<ref name="pmid18156035"/> Topical [[microbicide]]s that contain chemicals that directly inactivate the virus and block viral entry are being investigated.<ref name="pmid18156035"/> |
|||
===Antivirals=== |
===Antivirals=== |
||
| Line 168: | Line 166: | ||
===Pregnancy=== |
===Pregnancy=== |
||
The risk of transmission from mother to baby is highest if the mother becomes infected around the time of delivery (30% to 60%),<ref>{{cite journal|author=Brown ZA|title=The acquisition of herpes simplex virus during pregnancy|journal=N Engl J Med|volume=337|pages=509–15|year=1997|doi=10.1056/NEJM199708213370801|pmid=9262493|name-list-style=vanc|author2=Selke S|author3=Zeh J|display-authors=3|last4=Kopelman|first4=Jerome|last5=Maslow|first5=Arthur|last6=Ashley|first6=Rhoda L.|last7=Watts|first7=D. Heather|last8=Berry|first8=Sylvia|last9=Herd|first9=Millie|issue=8|doi-access=free}}</ref><ref>{{cite journal|vauthors=Brown ZA, Wald A, Morrow RA, Selke S, Zeh J, Corey L |title=Effect of serologic status and cesarean delivery on transmission rates of herpes simplex virus from mother to infant|journal=JAMA|volume=289|pages=203–09|year=2003|doi=10.1001/jama.289.2.203|pmid=12517231|issue=2|doi-access=free}}</ref> since insufficient time will have occurred for the generation and transfer of protective maternal antibodies before the birth of the child. In contrast, the risk falls to 3% if the infection is recurrent,<ref name=pmid1849612/> and is 1–3% if the woman is seropositive for both HSV-1 and HSV-2,<ref name=pmid1849612>{{cite journal |author=Brown ZA |title=Neonatal herpes simplex virus infection in relation to asymptomatic maternal infection at the time of labor |journal=N. Engl. J. Med. |volume=324 |issue=18 |pages=1247–52 |date=May 1991 |pmid=1849612 |doi= 10.1056/NEJM199105023241804 |name-list-style=vanc|author2=Benedetti J |author3=Ashley R |display-authors=3 |last4=Burchett |first4=Sandra |last5=Selke |first5=Stacy |last6=Berry |first6=Sylvia |last7=Vontver |first7=Louis A. |last8=Corey |first8=Lawrence}}</ref><ref name=Whitley_RJ>{{cite journal|vauthors=Whitley RJ, Kimberlin DW, Roizman B | title=Herpes simplex viruses| journal=Clin Infect Dis| year=1998| pages=541–53| volume=26| issue=3| pmid=9524821|doi=10.1086/514600| doi-access=free}}</ref> and is less than 1% if no lesions are visible.<ref name=pmid1849612/> Women seropositive for only one type of HSV are only half as likely to transmit HSV as infected seronegative mothers. To prevent neonatal infections, seronegative women are recommended to avoid unprotected oral-genital contact with an HSV-1-seropositive partner and conventional sex with a partner having a genital infection during the last trimester of pregnancy. Mothers infected with HSV are advised to avoid procedures that would cause trauma to the infant during birth (e.g. fetal scalp electrodes, forceps, and vacuum extractors) and, should lesions be present, to elect [[caesarean section]] to reduce exposure of the child to infected secretions in the birth canal.<ref name="pmid18156035"/> The use of antiviral treatments, such as aciclovir, given from the 36th week of pregnancy, limits HSV recurrence and shedding during childbirth, thereby reducing the need for caesarean section.<ref name="pmid18156035"/> |
The risk of transmission from mother to baby is highest if the mother becomes infected around the time of delivery (30% to 60%),<ref>{{cite journal|author=Brown ZA|title=The acquisition of herpes simplex virus during pregnancy|journal=N Engl J Med|volume=337|pages=509–15|year=1997|doi=10.1056/NEJM199708213370801|pmid=9262493|name-list-style=vanc|author2=Selke S|author3=Zeh J|display-authors=3|last4=Kopelman|first4=Jerome|last5=Maslow|first5=Arthur|last6=Ashley|first6=Rhoda L.|last7=Watts|first7=D. Heather|last8=Berry|first8=Sylvia|last9=Herd|first9=Millie|issue=8|doi-access=free}}</ref><ref>{{cite journal|vauthors=Brown ZA, Wald A, Morrow RA, Selke S, Zeh J, Corey L |title=Effect of serologic status and cesarean delivery on transmission rates of herpes simplex virus from mother to infant|journal=JAMA|volume=289|pages=203–09|year=2003|doi=10.1001/jama.289.2.203|pmid=12517231|issue=2|doi-access=free}}</ref> since insufficient time will have occurred for the generation and transfer of protective maternal antibodies before the birth of the child. In contrast, the risk falls to 3% if the infection is recurrent,<ref name=pmid1849612/> and is 1–3% if the woman is seropositive for both HSV-1 and HSV-2,<ref name=pmid1849612>{{cite journal |author=Brown ZA |title=Neonatal herpes simplex virus infection in relation to asymptomatic maternal infection at the time of labor |journal=N. Engl. J. Med. |volume=324 |issue=18 |pages=1247–52 |date=May 1991 |pmid=1849612 |doi= 10.1056/NEJM199105023241804 |name-list-style=vanc|author2=Benedetti J |author3=Ashley R |display-authors=3 |last4=Burchett |first4=Sandra |last5=Selke |first5=Stacy |last6=Berry |first6=Sylvia |last7=Vontver |first7=Louis A. |last8=Corey |first8=Lawrence|doi-access=free }}</ref><ref name=Whitley_RJ>{{cite journal|vauthors=Whitley RJ, Kimberlin DW, Roizman B | title=Herpes simplex viruses| journal=Clin Infect Dis| year=1998| pages=541–53| volume=26| issue=3| pmid=9524821|doi=10.1086/514600| doi-access=free}}</ref> and is less than 1% if no lesions are visible.<ref name=pmid1849612/> Women seropositive for only one type of HSV are only half as likely to transmit HSV as infected seronegative mothers. To prevent neonatal infections, seronegative women are recommended to avoid unprotected oral-genital contact with an HSV-1-seropositive partner and conventional sex with a partner having a genital infection during the last trimester of pregnancy. Mothers infected with HSV are advised to avoid procedures that would cause trauma to the infant during birth (e.g. fetal scalp electrodes, forceps, and vacuum extractors) and, should lesions be present, to elect [[caesarean section]] to reduce exposure of the child to infected secretions in the birth canal.<ref name="pmid18156035"/> The use of antiviral treatments, such as aciclovir, given from the 36th week of pregnancy, limits HSV recurrence and shedding during childbirth, thereby reducing the need for caesarean section.<ref name="pmid18156035"/> |
||
Aciclovir is the recommended antiviral for herpes suppressive therapy during the last months of pregnancy. The use of valaciclovir and famciclovir, while potentially improving compliance, have less-well-determined safety in pregnancy. |
Aciclovir is the recommended antiviral for herpes suppressive therapy during the last months of pregnancy. The use of valaciclovir and famciclovir, while potentially improving compliance, have less-well-determined safety in pregnancy. |
||
| Line 179: | Line 177: | ||
Several [[antiviral drug]]s are effective for treating herpes, including [[aciclovir]] (acyclovir), [[valaciclovir]], [[famciclovir]], and [[penciclovir]]. Aciclovir was the first discovered and is now available in [[generic medications|generic]].<ref>{{cite book|author = Robert L. LaFemina|title=Antiviral research : strategies in antiviral drug discovery|year=2009|publisher=ASM Press|location=Washington, DC|isbn=978-1-55581-439-7|pages=1|url=https://books.google.com/books?id=kyXjngXgU5YC&pg=PA1|url-status=live|archive-url=https://web.archive.org/web/20160502123637/https://books.google.com/books?id=kyXjngXgU5YC&pg=PA1|archive-date=2016-05-02}}</ref> Valaciclovir is also available as a generic<ref>{{cite book|last=Agrawal|first=Caroline A. Hastings, Joseph Torkildson, Anurag Kishor|title=Handbook of pediatric hematology and oncology : Children's Hospital & Research Center Oakland|publisher=Wiley-Blackwell|location=Chichester, West Sussex|isbn=978-0-470-67088-0|pages=360|url=https://books.google.com/books?id=H4t4PjtC730C&pg=PA360|edition=2nd|url-status=live|archive-url=https://web.archive.org/web/20160430224839/https://books.google.com/books?id=H4t4PjtC730C&pg=PA360|archive-date=2016-04-30|date=2012-04-30}}</ref> and is slightly more effective than aciclovir for reducing lesion healing time.<ref name="ChenXu2017">{{cite journal|last1=Chen|first1=Fangman|last2=Xu|first2=Hao|last3=Liu|first3=Jinli|last4=Cui|first4=Yuan|last5=Luo|first5=Xiaobo|last6=Zhou|first6=Yu|last7=Chen|first7=Qianming|last8=Jiang|first8=Lu|title=Efficacy and safety of nucleoside antiviral drugs for treatment of recurrent herpes labialis: a systematic review and meta-analysis|journal=Journal of Oral Pathology & Medicine|volume=46|issue=8|pages=561–568|year=2017|issn=0904-2512|doi=10.1111/jop.12534|pmid=27935123|s2cid=10391761}}</ref> |
Several [[antiviral drug]]s are effective for treating herpes, including [[aciclovir]] (acyclovir), [[valaciclovir]], [[famciclovir]], and [[penciclovir]]. Aciclovir was the first discovered and is now available in [[generic medications|generic]].<ref>{{cite book|author = Robert L. LaFemina|title=Antiviral research : strategies in antiviral drug discovery|year=2009|publisher=ASM Press|location=Washington, DC|isbn=978-1-55581-439-7|pages=1|url=https://books.google.com/books?id=kyXjngXgU5YC&pg=PA1|url-status=live|archive-url=https://web.archive.org/web/20160502123637/https://books.google.com/books?id=kyXjngXgU5YC&pg=PA1|archive-date=2016-05-02}}</ref> Valaciclovir is also available as a generic<ref>{{cite book|last=Agrawal|first=Caroline A. Hastings, Joseph Torkildson, Anurag Kishor|title=Handbook of pediatric hematology and oncology : Children's Hospital & Research Center Oakland|publisher=Wiley-Blackwell|location=Chichester, West Sussex|isbn=978-0-470-67088-0|pages=360|url=https://books.google.com/books?id=H4t4PjtC730C&pg=PA360|edition=2nd|url-status=live|archive-url=https://web.archive.org/web/20160430224839/https://books.google.com/books?id=H4t4PjtC730C&pg=PA360|archive-date=2016-04-30|date=2012-04-30}}</ref> and is slightly more effective than aciclovir for reducing lesion healing time.<ref name="ChenXu2017">{{cite journal|last1=Chen|first1=Fangman|last2=Xu|first2=Hao|last3=Liu|first3=Jinli|last4=Cui|first4=Yuan|last5=Luo|first5=Xiaobo|last6=Zhou|first6=Yu|last7=Chen|first7=Qianming|last8=Jiang|first8=Lu|title=Efficacy and safety of nucleoside antiviral drugs for treatment of recurrent herpes labialis: a systematic review and meta-analysis|journal=Journal of Oral Pathology & Medicine|volume=46|issue=8|pages=561–568|year=2017|issn=0904-2512|doi=10.1111/jop.12534|pmid=27935123|s2cid=10391761}}</ref> |
||
Evidence supports the use of aciclovir and valaciclovir in the treatment of herpes labialis<ref name=Best07>{{cite journal |vauthors=Chon T, Nguyen L, Elliott TC |title=Clinical inquiries. What are the best treatments for herpes labialis? |journal=J Fam Pract |volume=56 |issue=7 |pages=576–78 |date=July 2007 |pmid=17605952 }}</ref> as well as herpes infections in people with [[cancer]].<ref name="pmid19160295">{{cite journal | vauthors = Glenny AM, Fernandez Mauleffinch LM, Pavitt S, Walsh T | title = Interventions for the prevention and treatment of herpes simplex virus in patients being treated for cancer | journal = The Cochrane Database of Systematic Reviews | issue = 1 | pages = CD006706 | date = January 2009 | pmid = 19160295 | doi = 10.1002/14651858.CD006706.pub2}}</ref> The evidence to support the use of aciclovir in primary herpetic gingivostomatitis is weaker.<ref name="pmid18843726">{{cite journal | vauthors = Nasser M, Fedorowicz Z, Khoshnevisan MH, Shahiri Tabarestani M | title = Acyclovir for treating primary herpetic gingivostomatitis | journal = The Cochrane Database of Systematic Reviews | issue = 4 | pages = CD006700 | date = October 2008 | pmid = 18843726 | doi = 10.1002/14651858.CD006700.pub2| editor1-last = Nasser | editor1-first = Mona }} {{Retracted |
Evidence supports the use of aciclovir and valaciclovir in the treatment of herpes labialis<ref name=Best07>{{cite journal |vauthors=Chon T, Nguyen L, Elliott TC |title=Clinical inquiries. What are the best treatments for herpes labialis? |journal=J Fam Pract |volume=56 |issue=7 |pages=576–78 |date=July 2007 |pmid=17605952 }}</ref> as well as herpes infections in people with [[cancer]].<ref name="pmid19160295">{{cite journal | vauthors = Glenny AM, Fernandez Mauleffinch LM, Pavitt S, Walsh T | title = Interventions for the prevention and treatment of herpes simplex virus in patients being treated for cancer | journal = The Cochrane Database of Systematic Reviews | issue = 1 | pages = CD006706 | date = January 2009 | pmid = 19160295 | doi = 10.1002/14651858.CD006706.pub2}}</ref> The evidence to support the use of aciclovir in primary herpetic gingivostomatitis is weaker.<ref name="pmid18843726">{{cite journal | vauthors = Nasser M, Fedorowicz Z, Khoshnevisan MH, Shahiri Tabarestani M | title = Acyclovir for treating primary herpetic gingivostomatitis | journal = The Cochrane Database of Systematic Reviews | issue = 4 | pages = CD006700 | date = October 2008 | pmid = 18843726 | doi = 10.1002/14651858.CD006700.pub2| editor1-last = Nasser | editor1-first = Mona }} {{Retracted|doi=10.1002/14651858.CD006700.pub3|pmid=26784280|http://retractionwatch.com/2010/09/20/progressive-how-the-cochrane-library-handles-updates-in-progress/ ''Retraction Watch''}}</ref> |
||
===Topical=== |
===Topical=== |
||
| Line 202: | Line 200: | ||
Worldwide rates of either HSV-1 and/or HSV-2 are between 60 and 95% in adults.<ref name=Peds09/> HSV-1 is more common than HSV-2, with rates of both increasing as people age.<ref name=Peds09/> HSV-1 rates are between 70% and 80% in populations of low socioeconomic status and 40% to 60% in populations of improved socioeconomic status.<ref name=Peds09/> An estimated 536 million people or 16% of the population worldwide were infected with HSV-2 as of 2003 with greater rates among women and in those in the developing world.<ref name="Looker2008"/> Rates of infection are determined by the presence of [[antibodies]] against either [[viral species]].<ref name="pmid12353183">{{cite journal |vauthors=Smith JS, Robinson NJ |title=Age-specific prevalence of infection with herpes simplex virus types 2 and 1: a global review |journal=J. Infect. Dis. |volume=186 |issue=Suppl 1 |pages=S3–28 |year=2002 |pmid=12353183 |doi=10.1086/343739|doi-access=free }}</ref> |
Worldwide rates of either HSV-1 and/or HSV-2 are between 60 and 95% in adults.<ref name=Peds09/> HSV-1 is more common than HSV-2, with rates of both increasing as people age.<ref name=Peds09/> HSV-1 rates are between 70% and 80% in populations of low socioeconomic status and 40% to 60% in populations of improved socioeconomic status.<ref name=Peds09/> An estimated 536 million people or 16% of the population worldwide were infected with HSV-2 as of 2003 with greater rates among women and in those in the developing world.<ref name="Looker2008"/> Rates of infection are determined by the presence of [[antibodies]] against either [[viral species]].<ref name="pmid12353183">{{cite journal |vauthors=Smith JS, Robinson NJ |title=Age-specific prevalence of infection with herpes simplex virus types 2 and 1: a global review |journal=J. Infect. Dis. |volume=186 |issue=Suppl 1 |pages=S3–28 |year=2002 |pmid=12353183 |doi=10.1086/343739|doi-access=free }}</ref> |
||
In the [[United States|US]], 58% of the population is infected with HSV-1<ref name="JAMA. 2006;296:964-973.">{{cite journal|last=Xu|first=Fujie|author2=Fujie Xu|author3=Maya R. Sternberg|author4=Benny J. Kottiri|author5=Geraldine M. McQuillan|author6=Francis K. Lee|author7=Andre J. Nahmias|author8=Stuart M. Berman|author9=Lauri E. Markowitz|date=2006-10-23|title=Trends in Herpes Simplex Virus Type 1 and Type 2 Seroprevalence in the United States|journal=JAMA|volume=296|issue=8|pmid=16926356|doi=10.1001/jama.296.8.964|pages=964–73|doi-access= |
In the [[United States|US]], 58% of the population is infected with HSV-1<ref name="JAMA. 2006;296:964-973.">{{cite journal|last=Xu|first=Fujie|author2=Fujie Xu|author3=Maya R. Sternberg|author4=Benny J. Kottiri|author5=Geraldine M. McQuillan|author6=Francis K. Lee|author7=Andre J. Nahmias|author8=Stuart M. Berman|author9=Lauri E. Markowitz|date=2006-10-23|title=Trends in Herpes Simplex Virus Type 1 and Type 2 Seroprevalence in the United States|journal=JAMA|volume=296|issue=8|pmid=16926356|doi=10.1001/jama.296.8.964|pages=964–73|doi-access=}}</ref> and 16% are infected with HSV-2. Among those HSV-2-seropositive, only 19% were aware they were infected.<ref>{{cite journal|last=Xu|first=F|author2=MR Sternberg|author3=SL Gottlieb|author4=SM Berman|author5=LE Markowitz|display-authors=etal|title=Seroprevalence of Herpes Simplex Virus Type 2 Among Persons Aged 14–49 Years – United States, 2005–2008|journal=Morbidity and Mortality Weekly Report|date=23 April 2010|volume=59|issue=15|pages=456–59|pmid=20414188|url=https://www.cdc.gov/mmwr/preview/mmwrhtml/mm5915a3.htm|access-date=12 April 2011|url-status=live|archive-url=https://web.archive.org/web/20110625091920/http://www.cdc.gov/mmwr/preview/mmwrhtml/mm5915a3.htm|archive-date=25 June 2011}}</ref> During 2005–2008, the prevalence of HSV-2 was 39% in black people and 21% in women.<ref>{{cite web |url=https://www.cdc.gov/stdconference/2010/hsv2pressrelease.pdf |title=CDC Study Finds U.S. Herpes Rates Remain High |publisher=Center for Disease Control and Prevention |date=2010-03-09 |access-date=2012-02-19 |url-status=live |archive-url=https://web.archive.org/web/20160306090032/http://www.cdc.gov/stdconference/2010/hsv2pressrelease.pdf |archive-date=2016-03-06 }}</ref> |
||
The annual incidence in Canada of genital herpes due to HSV-1 and HSV-2 infection is not known (for a review of HSV-1/HSV-2 prevalence and incidence studies worldwide, see Smith and Robinson 2002). As many as one in seven Canadians aged 14 to 59 may be infected with herpes simplex type 2 virus<ref>{{Cite journal |last1=Rotermann |first1=Michelle |last2=Langlois |first2=Kellie A. |last3=Severini |first3=Alberto |last4=Totten |first4=Stephanie |date=2013-04-01 |title=Prevalence of Chlamydia trachomatis and herpes simplex virus type 2: Results from the 2009 to 2011 Canadian Health Measures Survey |journal=Health Reports |volume=24 |issue=4 |pages=10–15 |issn=1209-1367 |pmid=24258059}}</ref> and more than 90 per cent of them may be unaware of their status, a new study suggests.<ref>{{Cite news |url=http://news.nationalpost.com/health/herpes-virus-has-infected-nearly-one-in-five-canadians-over-age-35-most-unaware-they-have-it-study |title=Herpes virus has infected nearly one in five Canadians over age 35, most unaware they have it: study |newspaper=National Post |access-date=2017-02-20}}</ref> In the United States, it is estimated that about 1,640,000 HSV-2 seroconversions occur yearly (730,000 men and 910,000 women, or 8.4 per 1,000 persons).<ref>{{cite journal |author = Smith JS, Robinson NJ |year = 2002 |title = Age-specific prevalence of infection with herpes simplex virus types 2 and 1: a global review |journal = J Infect Dis |volume = 186 |issue = suppl 1 |pages = S3–28 |doi=10.1086/343739 |pmid = 12353183 |doi-access = free }}</ref> |
The annual incidence in Canada of genital herpes due to HSV-1 and HSV-2 infection is not known (for a review of HSV-1/HSV-2 prevalence and incidence studies worldwide, see Smith and Robinson 2002). As many as one in seven Canadians aged 14 to 59 may be infected with herpes simplex type 2 virus<ref>{{Cite journal |last1=Rotermann |first1=Michelle |last2=Langlois |first2=Kellie A. |last3=Severini |first3=Alberto |last4=Totten |first4=Stephanie |date=2013-04-01 |title=Prevalence of Chlamydia trachomatis and herpes simplex virus type 2: Results from the 2009 to 2011 Canadian Health Measures Survey |journal=Health Reports |volume=24 |issue=4 |pages=10–15 |issn=1209-1367 |pmid=24258059}}</ref> and more than 90 per cent of them may be unaware of their status, a new study suggests.<ref>{{Cite news |url=http://news.nationalpost.com/health/herpes-virus-has-infected-nearly-one-in-five-canadians-over-age-35-most-unaware-they-have-it-study |title=Herpes virus has infected nearly one in five Canadians over age 35, most unaware they have it: study |newspaper=National Post |access-date=2017-02-20}}</ref> In the United States, it is estimated that about 1,640,000 HSV-2 seroconversions occur yearly (730,000 men and 910,000 women, or 8.4 per 1,000 persons).<ref>{{cite journal |author = Smith JS, Robinson NJ |year = 2002 |title = Age-specific prevalence of infection with herpes simplex virus types 2 and 1: a global review |journal = J Infect Dis |volume = 186 |issue = suppl 1 |pages = S3–28 |doi=10.1086/343739 |pmid = 12353183 |doi-access = free }}</ref> |
||
| Line 213: | Line 211: | ||
== History == |
== History == |
||
Herpes originated and evolved in Africa and could be the result of a cross-species transmission event from gibbons, orangutans, or gorillas.<ref>{{Cite journal |last1=Wertheim |first1=Joel O. |last2=Smith |first2=Martin D. |last3=Smith |first3=Davey M. |last4=Scheffler |first4=Konrad |last5=Kosakovsky Pond |first5=Sergei L. |date=2014 |title=Evolutionary Origins of Human Herpes Simplex Viruses 1 and 2 |url=https://academic.oup.com/mbe/article-lookup/doi/10.1093/molbev/msu185 |journal=Molecular Biology and Evolution |language=en |volume=31 |issue=9 |pages=2356–2364 |doi=10.1093/molbev/msu185 |pmid=24916030 |issn=1537-1719|pmc=4137711 }}</ref> |
|||
| ⚫ | Herpes has been known for at least 2,000 years. Emperor [[Tiberius]] is said to have banned kissing in Rome for a time due to so many people having cold sores. In the 16th century ''[[Romeo and Juliet]]'', blisters "o'er ladies' lips" are mentioned. In the 18th century, it was so common among prostitutes that it was called "a vocational disease of women".<ref name="scarlet">{{cite news |url=http://www.time.com/time/magazine/article/0,9171,1715020,00.html |title=The New Scarlet Letter |author=John Leo |date=1982-08-02 |magazine=[[Time (magazine)|Time]] |archive-url=https://web.archive.org/web/20100202153257/http://www.time.com/time/magazine/article/0%2C9171%2C1715020%2C00.html |archive-date=2010-02-02 |url-status=dead}}</ref> The term 'herpes simplex' appeared in [[Richard Boulton]]'s ''A System of Rational and Practical Chirurgery'' in 1713, where the terms 'herpes miliaris' and 'herpes exedens' also appeared. Herpes was not found to be a virus until the 1940s.<ref name=scarlet /> |
||
| ⚫ | Herpes has been known for at least 2,000 years. Emperor [[Tiberius]] is said to have banned kissing in Rome for a time due to so many people having cold sores. In the 16th century ''[[Romeo and Juliet]]'', blisters "o'er ladies' lips" are mentioned. In the 18th century, it was so common among prostitutes that it was called "a vocational disease of women".<ref name="scarlet">{{cite news |url=http://www.time.com/time/magazine/article/0,9171,1715020,00.html |title=The New Scarlet Letter |author=John Leo |date=1982-08-02 |magazine=[[Time (magazine)|Time]] |archive-url=https://web.archive.org/web/20100202153257/http://www.time.com/time/magazine/article/0%2C9171%2C1715020%2C00.html |archive-date=2010-02-02 |url-status=dead}}</ref> The term 'herpes simplex' appeared in [[Richard Boulton]]'s ''A System of Rational and Practical Chirurgery'' in 1713, where the terms 'herpes miliaris' and 'herpes exedens' also appeared. Herpes was not found to be a virus until the 1940s.<ref name="scarlet" /> |
||
Herpes antiviral therapy began in the early 1960s with the experimental use of medications that interfered with viral replication called [[deoxyribonucleic acid]] (DNA) inhibitors. The original use was against normally fatal or debilitating illnesses such as adult encephalitis,<ref name="pmid4790599">{{cite journal|author=Chow AW|title=Cytosine Arabinoside Therapy for Herpes Simplex Encephalitis – Clinical Experience with Six Patients|journal=Antimicrob. Agents Chemother.|volume=3|issue=3|pages=412–17|date=March 1973|pmid=4790599|pmc=444424|doi=10.1128/aac.3.3.412|name-list-style=vanc|author2=Roland A|author3=Fiala M|display-authors=3|last4=Hryniuk|first4=W|last5=Weil|first5=ML|last6=Geme Jr|first6=JS|last7=Guze|first7=LB}}</ref> keratitis,<ref name="pmid14454441">{{cite journal | vauthors = Kaufman HE, Howard GM| title = Therapy of experimental herpes simplex keratitis | journal = Investigative Ophthalmology | volume = 1 | pages = 561–4 | date = August 1962 | pmid = 14454441 }}</ref> in immunocompromised (transplant) patients,<ref name="pmid180198">{{cite journal|vauthors=Ch'ien LT, Whitley RJ, Alford CA, Galasso GJ |title=Adenine arabinoside for therapy of herpes zoster in immunosuppressed patients: preliminary results of a collaborative study|journal=J. Infect. Dis.|volume=133|issue=Suppl|pages=A184–91|date=June 1976|pmid=180198|doi=10.1093/infdis/133.supplement_2.a184}}</ref> or [[disseminated herpes zoster]] (also known as disseminated shingles).<ref name="pmid5352659">{{cite journal|vauthors=McKelvey EM, Kwaan HC|title=Cytosine arabinoside therapy for disseminated herpes zoster in a patient with IgG pyroglobulinemia|journal=Blood|volume=34|issue=5|pages=706–11|date=November 1969|pmid=5352659|doi=10.1182/blood.V34.5.706.706|doi-access=free}}</ref> The original compounds used were 5-iodo-2'-deoxyuridine, AKA idoxuridine, IUdR, or(IDU) and 1-β-D-arabinofuranosylcytosine or ara-C,<ref name="pmid4364937">{{cite journal|vauthors=Fiala M, Chow A, Guze LB |title=Susceptibility of Herpesviruses to Cytosine Arabinoside: Standardization of Susceptibility Test Procedure and Relative Resistance of Herpes Simplex Type 2 Strains|journal=Antimicrob. Agents Chemother.|volume=1|issue=4|pages=354–57|date=April 1972|pmid=4364937|pmc=444221|doi=10.1128/aac.1.4.354}}</ref> later marketed under the name cytosar or cytarabine. The usage expanded to include topical treatment of herpes simplex,<ref name="pmid6598">{{cite journal|author=Allen LB|title=Effect of 9-beta-D-arabinofuranosylhypoxanthine 5'-monophosphate on genital lesions and encephalitis induced by Herpesvirus hominis type 2 in female mice|journal=J. Infect. Dis.|volume=133|issue= Suppl|pages=A178–83|date=June 1976|pmid=6598|name-list-style=vanc|author2=Hintz OJ|author3=Wolf SM|display-authors=3|last4=Huffman|first4=JH|last5=Simon|first5=LN|last6=Robins|first6=RK|last7=Sidwell|first7=RW|doi=10.1093/infdis/133.supplement_2.a178}}</ref> zoster, and varicella.<ref name="pmid4190397">{{cite journal|author=Juel-Jensen BE|title=Varicella and cytosine arabinoside|journal=Lancet|volume=1|issue=7646|page=572|date=March 1970|pmid=4190397|doi=10.1016/S0140-6736(70)90815-9}}</ref> Some trials combined different antivirals with differing results.<ref name="pmid4790599"/> The introduction of 9-β-D-arabinofuranosyladenine, (ara-A or vidarabine), considerably less toxic than ara-C, in the mid-1970s, heralded the way for the beginning of regular neonatal antiviral treatment. Vidarabine was the first systemically administered antiviral medication with activity against HSV for which therapeutic efficacy outweighed toxicity for the management of life-threatening HSV disease. Intravenous vidarabine was licensed for use by the [[U.S. Food and Drug Administration]] in 1977. Other experimental antivirals of that period included: heparin,<ref name="pmid4289440">{{cite journal|vauthors=Nahmias AJ, Kibrick S |title=Inhibitory Effect of Heparin on Herpes Simplex Virus|journal=J. Bacteriol.|volume=87|issue=5|pages=1060–66|date=May 1964|pmid=4289440|pmc=277146|doi=10.1128/JB.87.5.1060-1066.1964}}</ref> trifluorothymidine (TFT),<ref name="pmid4790562">{{cite journal|vauthors=Allen LB, Sidwell RW |title=Target-Organ Treatment of Neurotropic Virus Diseases: Efficacy as a Chemotherapy Tool and Comparison of Activity of Adenine Arabinoside, Cytosine Arabinoside, Idoxuridine, and Trifluorothymidine|journal=Antimicrob. Agents Chemother.|volume=2|issue=3|pages=229–33|date=September 1972|pmid=4790562|pmc=444296|doi=10.1128/aac.2.3.229}}</ref> Ribivarin,<ref name="pmid212976">{{cite journal|vauthors=Allen LB, Wolf SM, Hintz CJ, Huffman JH, Sidwell RW |title=Effect of ribavirin on Type 2 Herpesvirus hominis (HVH/2) ''in vitro'' and ''in vivo''|journal=Annals of the New York Academy of Sciences|volume=284|issue=1|pages=247–53|date=March 1977|pmid=212976|doi=10.1111/j.1749-6632.1977.tb21957.x|bibcode=1977NYASA.284..247A|s2cid=12159958}}</ref> interferon,<ref name="pmid4404669">{{cite journal|vauthors=Allen LB, Cochran KW |title=Target-Organ Treatment of Neurotropic Virus Disease with Interferon Inducers|journal=Infection and Immunity|volume=6|issue=5|pages=819–23|date=November 1972|pmid=4404669|pmc=422616|doi=10.1128/IAI.6.5.819-823.1972}}</ref> Virazole,<ref name="pmid4340949">{{cite journal|vauthors=Sidwell RW, Huffman JH, Khare GP, Allen LB, Witkowski JT, Robins RK |title=Broad-spectrum antiviral activity of Virazole: 1-beta-D-ribofuranosyl-1,2,4-triazole-3-carboxamide|journal=Science|volume=177|issue=4050|pages=705–06|date=August 1972|pmid=4340949|doi=10.1126/science.177.4050.705|bibcode=1972Sci...177..705S|s2cid=43106875}}</ref> and 5-methoxymethyl-2'-deoxyuridine (MMUdR).<ref name="pmid1239978">{{cite journal|vauthors=Babiuk LA, Meldrum B, Gupta VS, Rouse BT |title=Comparison of the Antiviral Effects of 5-Methoxymethyl-deoxyuridine with 5-Iododeoxyuridine, Cytosine Arabinoside, and Adenine Arabinoside|journal=Antimicrob. Agents Chemother.|volume=8|issue=6|pages=643–50|date=December 1975|pmid=1239978|pmc=429441|doi=10.1128/aac.8.6.643}}</ref> The introduction of 9-(2-hydroxyethoxymethyl)guanine, AKA aciclovir, in the late 1970s<ref name="pmid91931">{{cite journal|vauthors=O'Meara A, Deasy PF, Hillary IB, Bridgen WD |title=Acyclovir for treatment of mucocutaneous herpes infection in a child with leukaemia|journal=Lancet|volume=2|issue=8153|page=1196|date=December 1979|pmid=91931|doi=10.1016/S0140-6736(79)92428-0|s2cid=40775996}}</ref> raised antiviral treatment another notch and led to vidarabine vs. aciclovir trials in the late 1980s.<ref name="pmid1988829">{{cite journal|author=Whitley R|title=A controlled trial comparing vidarabine with acyclovir in neonatal herpes simplex virus infection. Infectious Diseases Collaborative Antiviral Study Group|journal=N. Engl. J. Med.|volume=324|issue=7|pages=444–49|date=February 1991|pmid=1988829|doi=10.1056/NEJM199102143240703|name-list-style=vanc|author2=Arvin A|author3=Prober C|display-authors=3|last4=Burchett|first4=Sandra|last5=Corey|first5=Lawrence|last6=Powell|first6=Dwight|last7=Plotkin|first7=Stanley|last8=Starr|first8=Stuart|last9=Alford|first9=Charles|doi-access=free}}</ref> The lower toxicity and ease of administration over vidarabine has led to aciclovir becoming the drug of choice for herpes treatment after it was licensed by the FDA in 1998.<ref name="pmid11483782">{{cite journal|author=Kimberlin DW|title=Safety and efficacy of high-dose intravenous acyclovir in the management of neonatal herpes simplex virus infections|journal=Pediatrics|volume=108|issue=2|pages=230–38|date=August 2001|pmid=11483782|doi=10.1542/peds.108.2.230|url=http://pediatrics.aappublications.org/cgi/pmidlookup?view=long&pmid=11483782|name-list-style=vanc|author2=Lin CY|author3=Jacobs RF|display-authors=3|last4=Powell|first4=D. A.|last5=Corey|first5=L.|last6=Gruber|first6=W. C.|last7=Rathore|first7=M.|last8=Bradley|first8=J. S.|last9=Diaz|first9=P. S.}}</ref> Another advantage in the treatment of neonatal herpes included greater reductions in mortality and morbidity with increased dosages, which did not occur when compared with increased dosages of vidarabine.<ref name="pmid11483782"/> However, aciclovir seems to inhibit antibody response, and newborns on aciclovir antiviral treatment experienced a slower rise in antibody titer than those on vidarabine.<ref name="pmid11483782"/> |
Herpes antiviral therapy began in the early 1960s with the experimental use of medications that interfered with viral replication called [[deoxyribonucleic acid]] (DNA) inhibitors. The original use was against normally fatal or debilitating illnesses such as adult encephalitis,<ref name="pmid4790599">{{cite journal|author=Chow AW|title=Cytosine Arabinoside Therapy for Herpes Simplex Encephalitis – Clinical Experience with Six Patients|journal=Antimicrob. Agents Chemother.|volume=3|issue=3|pages=412–17|date=March 1973|pmid=4790599|pmc=444424|doi=10.1128/aac.3.3.412|name-list-style=vanc|author2=Roland A|author3=Fiala M|display-authors=3|last4=Hryniuk|first4=W|last5=Weil|first5=ML|last6=Geme Jr|first6=JS|last7=Guze|first7=LB}}</ref> keratitis,<ref name="pmid14454441">{{cite journal | vauthors = Kaufman HE, Howard GM| title = Therapy of experimental herpes simplex keratitis | journal = Investigative Ophthalmology | volume = 1 | pages = 561–4 | date = August 1962 | pmid = 14454441 }}</ref> in immunocompromised (transplant) patients,<ref name="pmid180198">{{cite journal|vauthors=Ch'ien LT, Whitley RJ, Alford CA, Galasso GJ |title=Adenine arabinoside for therapy of herpes zoster in immunosuppressed patients: preliminary results of a collaborative study|journal=J. Infect. Dis.|volume=133|issue=Suppl|pages=A184–91|date=June 1976|pmid=180198|doi=10.1093/infdis/133.supplement_2.a184}}</ref> or [[disseminated herpes zoster]] (also known as disseminated shingles).<ref name="pmid5352659">{{cite journal|vauthors=McKelvey EM, Kwaan HC|title=Cytosine arabinoside therapy for disseminated herpes zoster in a patient with IgG pyroglobulinemia|journal=Blood|volume=34|issue=5|pages=706–11|date=November 1969|pmid=5352659|doi=10.1182/blood.V34.5.706.706|doi-access=free}}</ref> The original compounds used were 5-iodo-2'-deoxyuridine, AKA idoxuridine, IUdR, or(IDU) and 1-β-D-arabinofuranosylcytosine or ara-C,<ref name="pmid4364937">{{cite journal|vauthors=Fiala M, Chow A, Guze LB |title=Susceptibility of Herpesviruses to Cytosine Arabinoside: Standardization of Susceptibility Test Procedure and Relative Resistance of Herpes Simplex Type 2 Strains|journal=Antimicrob. Agents Chemother.|volume=1|issue=4|pages=354–57|date=April 1972|pmid=4364937|pmc=444221|doi=10.1128/aac.1.4.354}}</ref> later marketed under the name cytosar or cytarabine. The usage expanded to include topical treatment of herpes simplex,<ref name="pmid6598">{{cite journal|author=Allen LB|title=Effect of 9-beta-D-arabinofuranosylhypoxanthine 5'-monophosphate on genital lesions and encephalitis induced by Herpesvirus hominis type 2 in female mice|journal=J. Infect. Dis.|volume=133|issue= Suppl|pages=A178–83|date=June 1976|pmid=6598|name-list-style=vanc|author2=Hintz OJ|author3=Wolf SM|display-authors=3|last4=Huffman|first4=JH|last5=Simon|first5=LN|last6=Robins|first6=RK|last7=Sidwell|first7=RW|doi=10.1093/infdis/133.supplement_2.a178}}</ref> zoster, and varicella.<ref name="pmid4190397">{{cite journal|author=Juel-Jensen BE|title=Varicella and cytosine arabinoside|journal=Lancet|volume=1|issue=7646|page=572|date=March 1970|pmid=4190397|doi=10.1016/S0140-6736(70)90815-9}}</ref> Some trials combined different antivirals with differing results.<ref name="pmid4790599"/> The introduction of 9-β-D-arabinofuranosyladenine, (ara-A or vidarabine), considerably less toxic than ara-C, in the mid-1970s, heralded the way for the beginning of regular neonatal antiviral treatment. Vidarabine was the first systemically administered antiviral medication with activity against HSV for which therapeutic efficacy outweighed toxicity for the management of life-threatening HSV disease. Intravenous vidarabine was licensed for use by the [[U.S. Food and Drug Administration]] in 1977. Other experimental antivirals of that period included: heparin,<ref name="pmid4289440">{{cite journal|vauthors=Nahmias AJ, Kibrick S |title=Inhibitory Effect of Heparin on Herpes Simplex Virus|journal=J. Bacteriol.|volume=87|issue=5|pages=1060–66|date=May 1964|pmid=4289440|pmc=277146|doi=10.1128/JB.87.5.1060-1066.1964}}</ref> trifluorothymidine (TFT),<ref name="pmid4790562">{{cite journal|vauthors=Allen LB, Sidwell RW |title=Target-Organ Treatment of Neurotropic Virus Diseases: Efficacy as a Chemotherapy Tool and Comparison of Activity of Adenine Arabinoside, Cytosine Arabinoside, Idoxuridine, and Trifluorothymidine|journal=Antimicrob. Agents Chemother.|volume=2|issue=3|pages=229–33|date=September 1972|pmid=4790562|pmc=444296|doi=10.1128/aac.2.3.229}}</ref> Ribivarin,<ref name="pmid212976">{{cite journal|vauthors=Allen LB, Wolf SM, Hintz CJ, Huffman JH, Sidwell RW |title=Effect of ribavirin on Type 2 Herpesvirus hominis (HVH/2) ''in vitro'' and ''in vivo''|journal=Annals of the New York Academy of Sciences|volume=284|issue=1|pages=247–53|date=March 1977|pmid=212976|doi=10.1111/j.1749-6632.1977.tb21957.x|bibcode=1977NYASA.284..247A|s2cid=12159958}}</ref> interferon,<ref name="pmid4404669">{{cite journal|vauthors=Allen LB, Cochran KW |title=Target-Organ Treatment of Neurotropic Virus Disease with Interferon Inducers|journal=Infection and Immunity|volume=6|issue=5|pages=819–23|date=November 1972|pmid=4404669|pmc=422616|doi=10.1128/IAI.6.5.819-823.1972}}</ref> Virazole,<ref name="pmid4340949">{{cite journal|vauthors=Sidwell RW, Huffman JH, Khare GP, Allen LB, Witkowski JT, Robins RK |title=Broad-spectrum antiviral activity of Virazole: 1-beta-D-ribofuranosyl-1,2,4-triazole-3-carboxamide|journal=Science|volume=177|issue=4050|pages=705–06|date=August 1972|pmid=4340949|doi=10.1126/science.177.4050.705|bibcode=1972Sci...177..705S|s2cid=43106875}}</ref> and 5-methoxymethyl-2'-deoxyuridine (MMUdR).<ref name="pmid1239978">{{cite journal|vauthors=Babiuk LA, Meldrum B, Gupta VS, Rouse BT |title=Comparison of the Antiviral Effects of 5-Methoxymethyl-deoxyuridine with 5-Iododeoxyuridine, Cytosine Arabinoside, and Adenine Arabinoside|journal=Antimicrob. Agents Chemother.|volume=8|issue=6|pages=643–50|date=December 1975|pmid=1239978|pmc=429441|doi=10.1128/aac.8.6.643}}</ref> The introduction of 9-(2-hydroxyethoxymethyl)guanine, AKA aciclovir, in the late 1970s<ref name="pmid91931">{{cite journal|vauthors=O'Meara A, Deasy PF, Hillary IB, Bridgen WD |title=Acyclovir for treatment of mucocutaneous herpes infection in a child with leukaemia|journal=Lancet|volume=2|issue=8153|page=1196|date=December 1979|pmid=91931|doi=10.1016/S0140-6736(79)92428-0|s2cid=40775996}}</ref> raised antiviral treatment another notch and led to vidarabine vs. aciclovir trials in the late 1980s.<ref name="pmid1988829">{{cite journal|author=Whitley R|title=A controlled trial comparing vidarabine with acyclovir in neonatal herpes simplex virus infection. Infectious Diseases Collaborative Antiviral Study Group|journal=N. Engl. J. Med.|volume=324|issue=7|pages=444–49|date=February 1991|pmid=1988829|doi=10.1056/NEJM199102143240703|name-list-style=vanc|author2=Arvin A|author3=Prober C|display-authors=3|last4=Burchett|first4=Sandra|last5=Corey|first5=Lawrence|last6=Powell|first6=Dwight|last7=Plotkin|first7=Stanley|last8=Starr|first8=Stuart|last9=Alford|first9=Charles|doi-access=free}}</ref> The lower toxicity and ease of administration over vidarabine has led to aciclovir becoming the drug of choice for herpes treatment after it was licensed by the FDA in 1998.<ref name="pmid11483782">{{cite journal|author=Kimberlin DW|title=Safety and efficacy of high-dose intravenous acyclovir in the management of neonatal herpes simplex virus infections|journal=Pediatrics|volume=108|issue=2|pages=230–38|date=August 2001|pmid=11483782|doi=10.1542/peds.108.2.230|url=http://pediatrics.aappublications.org/cgi/pmidlookup?view=long&pmid=11483782|name-list-style=vanc|author2=Lin CY|author3=Jacobs RF|display-authors=3|last4=Powell|first4=D. A.|last5=Corey|first5=L.|last6=Gruber|first6=W. C.|last7=Rathore|first7=M.|last8=Bradley|first8=J. S.|last9=Diaz|first9=P. S.}}</ref> Another advantage in the treatment of neonatal herpes included greater reductions in mortality and morbidity with increased dosages, which did not occur when compared with increased dosages of vidarabine.<ref name="pmid11483782"/> However, aciclovir seems to inhibit antibody response, and newborns on aciclovir antiviral treatment experienced a slower rise in antibody titer than those on vidarabine.<ref name="pmid11483782"/> |
||
== Society and culture == |
== Society and culture == |
||
Some people experience negative feelings related to the condition following diagnosis, in particular, if they have acquired the genital form of the disease. Feelings can include [[Depression (mood)|depression]], fear of rejection, feelings of [[Solitude|isolation]], fear of being found out, and self-destructive feelings.<ref name="Vezina">{{cite journal| vauthors=Vezina C, Steben M| title=Genital Herpes: Psychosexual Impacts and Counselling| journal=The Canadian Journal of CME| year=2001| pages=125–34| issue=June| url=http://www.stacommunications.com/journals/cme/images/cmepdf/june01/hsv.pdf| url-status=dead| archive-url=https://web.archive.org/web/20081216222217/http://www.stacommunications.com/journals/cme/images/cmepdf/june01/hsv.pdf| archive-date=2008-12-16| access-date=2008-09-10}}</ref> [[Herpes support groups]] have been formed in the United States and the United Kingdom, providing information about herpes and running message forums and dating websites for affected people. People with the herpes virus are often hesitant to divulge to other people, including friends and family, that they are infected. This is especially true of new or potential sexual partners whom they consider casual.<ref name="Green">{{cite journal|last=Green|first=J|author2=Ferrier, S |author3=Kocsis, A |author4=Shadrick, J |author5=Ukoumunne, OC |author6=Murphy, S |author7= Hetherton, J |title=Determinants of disclosure of genital herpes to partners.|journal=Sexually Transmitted Infections|date=February 2003|volume=79|issue=1|pages=42–44|pmid=12576613|pmc=1744583|doi=10.1136/sti.79.1.42}}</ref> |
Some people experience negative feelings related to the condition following diagnosis, in particular, if they have acquired the genital form of the disease. Feelings can include [[Depression (mood)|depression]], fear of rejection, feelings of [[Solitude|isolation]], fear of being found out, and self-destructive feelings.<ref name="Vezina">{{cite journal| vauthors=Vezina C, Steben M| title=Genital Herpes: Psychosexual Impacts and Counselling| journal=The Canadian Journal of CME| year=2001| pages=125–34| issue=June| url=http://www.stacommunications.com/journals/cme/images/cmepdf/june01/hsv.pdf| url-status=dead| archive-url=https://web.archive.org/web/20081216222217/http://www.stacommunications.com/journals/cme/images/cmepdf/june01/hsv.pdf| archive-date=2008-12-16| access-date=2008-09-10}}</ref> [[Herpes support groups]] have been formed in the United States and the United Kingdom, providing information about herpes and running message forums and dating websites for affected people. People with the herpes virus are often hesitant to divulge to other people, including friends and family, that they are infected. This is especially true of new or potential sexual partners whom they consider casual.<ref name="Green">{{cite journal|last=Green|first=J|author2=Ferrier, S |author3=Kocsis, A |author4=Shadrick, J |author5=Ukoumunne, OC |author6=Murphy, S |author7= Hetherton, J |title=Determinants of disclosure of genital herpes to partners.|journal=Sexually Transmitted Infections|date=February 2003|volume=79|issue=1|pages=42–44|pmid=12576613|pmc=1744583|doi=10.1136/sti.79.1.42}}</ref> |
||
| Line 228: | Line 228: | ||
==== UK ==== |
==== UK ==== |
||
| ⚫ | In the UK, the Herpes Association (now the [[Herpes Viruses Association]]) was started in 1982, becoming a registered charity with a Department of Health grant in 1985. The charity started as a string of local group meetings before acquiring an office and a national spread.<ref>{{Cite web |url=http://www.hva.org.uk |title=Helping You With Herpes |publisher=Herpes Viruses Association |access-date=2017-02-20 |url-status=live |archive-url=https://web.archive.org/web/20150726180701/http://www.hva.org.uk/|archive-date=2015-07-26}}</ref> |
||
| ⚫ | In the UK, the Herpes Association (now the [[Herpes Viruses Association]]) was started in 1982, becoming a registered charity with a |
||
==Research== |
==Research== |
||
{{Main|Herpes simplex research}} |
{{Main|Herpes simplex research}} |
||
Research has gone into vaccines for both prevention and treatment of herpes infections. |
Research has gone into vaccines for both prevention and treatment of herpes infections. |
||
As of October 2022, the U.S. [[Food and Drug Administration|FDA]] have not approved a vaccine for herpes.<ref>{{Cite web |title=mRNA-1608 Herpes Vaccine |url=https://www.precisionvaccinations.com/vaccines/mrna-1608-herpes-vaccine |access-date=2022-12-11 |website=www.precisionvaccinations.com |language=en-US}}</ref> However, there are herpes vaccines currently in clinical trials, such as [[Moderna]] mRNA-1608.<ref>{{Cite web |date=2022-09-27 |title=Can herpes kill you? - Technology Org |url=https://www.technology.org/2022/09/27/can-herpes-kill-you/ |access-date=2022-12-11 |website=www.technology.org |language=en-US}}</ref> Unsuccessful clinical trials have been conducted for some glycoprotein subunit vaccines.{{citation needed|date=May 2019}} As of 2017, the future pipeline includes several promising replication-incompetent vaccine proposals while two replication-competent (live-attenuated) HSV vaccine are undergoing human testing.{{citation needed|date=May 2019}} |
As of October 2022, the U.S. [[Food and Drug Administration|FDA]] have not approved a vaccine for herpes.<ref>{{Cite web |title=mRNA-1608 Herpes Vaccine |url=https://www.precisionvaccinations.com/vaccines/mrna-1608-herpes-vaccine |access-date=2022-12-11 |website=www.precisionvaccinations.com |language=en-US}}</ref> However, there are herpes vaccines currently in clinical trials, such as [[Moderna]] mRNA-1608.<ref>{{Cite web |date=2022-09-27 |title=Can herpes kill you? - Technology Org |url=https://www.technology.org/2022/09/27/can-herpes-kill-you/ |access-date=2022-12-11 |website=www.technology.org |language=en-US}}</ref> Unsuccessful clinical trials have been conducted for some glycoprotein subunit vaccines.{{citation needed|date=May 2019}} As of 2017, the future pipeline includes several promising replication-incompetent vaccine proposals while two replication-competent (live-attenuated) HSV vaccine are undergoing human testing.{{citation needed|date=May 2019}} |
||
| Line 244: | Line 243: | ||
==External links== |
==External links== |
||
{{Commons category|Herpes simplex}} |
{{Commons category|Herpes simplex}} |
||
<!--BEFORE inserting new links here you should first post it to the talk page, otherwise your edit is likely to be reverted--> |
<!-- BEFORE inserting new links here you should first post it to the talk page, otherwise your edit is likely to be reverted. --> |
||
* {{curlie|Health/Conditions_and_Diseases/Infectious_Diseases/Viral/Herpes/}} |
|||
{{Medical condition classification and resources |
{{Medical condition classification and resources |
||
| DiseasesDB = 5841 |
| DiseasesDB = 5841 |
||
| DiseasesDB_mult = {{DiseasesDB2|33021}} |
| DiseasesDB_mult = {{DiseasesDB2|33021}} |
||
| ICD11 = {{ICD11|1F00}} |
|||
| ICD10 = {{ICD10|A|60||a|50}}, {{ICD10|B|00||b|00}}, {{ICD10|G|05|1|g|00}}, {{ICD10|P|35|2|p|35}} |
| ICD10 = {{ICD10|A|60||a|50}}, {{ICD10|B|00||b|00}}, {{ICD10|G|05|1|g|00}}, {{ICD10|P|35|2|p|35}} |
||
| ICD9 = {{ICD9|054.0}}, {{ICD9|054.1}}, {{ICD9|054.2}}, {{ICD9|054.3}}, {{ICD9|771.2}} |
| ICD9 = {{ICD9|054.0}}, {{ICD9|054.1}}, {{ICD9|054.2}}, {{ICD9|054.3}}, {{ICD9|771.2}} |
||
| Line 270: | Line 269: | ||
[[Category:Conditions of the mucous membranes]] |
[[Category:Conditions of the mucous membranes]] |
||
[[Category:Herpes simplex virus–associated diseases]] |
[[Category:Herpes simplex virus–associated diseases]] |
||
| ⚫ | |||
[[Category:Sexually transmitted diseases and infections]] |
[[Category:Sexually transmitted diseases and infections]] |
||
| ⚫ | |||
[[Category:Viral diseases]] |
[[Category:Viral diseases]] |
||
| ⚫ | |||
[[Category:Wikipedia emergency medicine articles ready to translate]] |
[[Category:Wikipedia emergency medicine articles ready to translate]] |
||
| ⚫ | |||
Latest revision as of 17:14, 28 November 2024
| Herpes | |
|---|---|
| Other names | Herpes simplex |
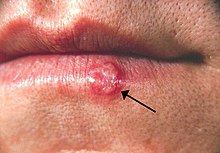 | |
| Oral herpes of the lower lip. Note the blisters in a group marked by an arrow. | |
| Pronunciation | |
| Specialty | Infectious disease |
| Symptoms | Blisters that break open and form small ulcers, fever, swollen lymph nodes[1] |
| Duration | 2–4 weeks[1] |
| Causes | Herpes simplex virus spread by direct contact[1] |
| Risk factors | Decreased immune function, stress, sunlight[2][3] |
| Diagnostic method | Based on symptoms, PCR, viral culture[1][2] |
| Medication | Aciclovir, valaciclovir, paracetamol (acetaminophen), topical lidocaine[1][2] |
| Frequency | 60–95% (adults)[4] |
Herpes simplex, often known simply as herpes, is a viral infection caused by the herpes simplex virus.[5] Herpes infections are categorized by the area of the body that is infected. The two major types of herpes are oral herpes and genital herpes, though other forms also exist.
Oral herpes involves the face or mouth. It may result in small blisters in groups, often called cold sores or fever blisters, or may just cause a sore throat.[2][6] Genital herpes involves the genitalia. It may have minimal symptoms or form blisters that break open and result in small ulcers.[1] These typically heal over two to four weeks.[1] Tingling or shooting pains may occur before the blisters appear.[1]
Herpes cycles between periods of active disease followed by periods without symptoms.[1] The first episode is often more severe and may be associated with fever, muscle pains, swollen lymph nodes and headaches.[1] Over time, episodes of active disease decrease in frequency and severity.[1]
Herpetic whitlow typically involves the fingers or thumb,[7] herpes simplex keratitis involves the eye,[8] herpesviral encephalitis involves the brain,[9] and neonatal herpes involves any part of the body of a newborn, among others.[10]
There are two types of herpes simplex virus, type 1 (HSV-1) and type 2 (HSV-2).[1] HSV-1 more commonly causes infections around the mouth while HSV-2 more commonly causes genital infections.[2] They are transmitted by direct contact with body fluids or lesions of an infected individual.[1] Transmission may still occur when symptoms are not present.[1] Genital herpes is classified as a sexually transmitted infection.[1] It may be spread to an infant during childbirth.[1] After infection, the viruses are transported along sensory nerves to the nerve cell bodies, where they reside lifelong.[2] Causes of recurrence may include: decreased immune function, stress, and sunlight exposure.[2][3] Oral and genital herpes is usually diagnosed based on the presenting symptoms.[2] The diagnosis may be confirmed by viral culture or detecting herpes DNA in fluid from blisters.[1] Testing the blood for antibodies against the virus can confirm a previous infection but will be negative in new infections.[1]
The most effective method of avoiding genital infections is by avoiding vaginal, oral, manual, and anal sex.[1][11] Condom use decreases the risk.[1] Daily antiviral medication taken by someone who has the infection can also reduce spread.[1] There is no available vaccine[1] and once infected, there is no cure.[1] Paracetamol (acetaminophen) and topical lidocaine may be used to help with the symptoms.[2] Treatments with antiviral medication such as aciclovir or valaciclovir can lessen the severity of symptomatic episodes.[1][2]
Worldwide rates of either HSV-1 or HSV-2 are between 60% and 95% in adults.[4] HSV-1 is usually acquired during childhood.[1] Since there is no cure for either HSV-1 or HSV-2, rates of both inherently increase as people age.[4] Rates of HSV-1 are between 70% and 80% in populations of low socioeconomic status and 40% to 60% in populations of improved socioeconomic status.[4] An estimated 536 million people worldwide (16% of the population) were infected with HSV-2 as of 2003 with greater rates among women and those in the developing world.[12] Most people with HSV-2 do not realize that they are infected.[1]
Etymology
The name is from Ancient Greek: ἕρπης herpēs, which is related to the meaning 'to creep', referring to spreading blisters.[13] The name does not refer to latency.[14]
Signs and symptoms

HSV infection causes several distinct medical disorders. Common infection of the skin or mucosa may affect the face and mouth (orofacial herpes), genitalia (genital herpes), or hands (herpetic whitlow). More serious disorders occur when the virus infects and damages the eye (herpes keratitis), or invades the central nervous system, damaging the brain (herpes encephalitis). People with immature or suppressed immune systems, such as newborns, transplant recipients, or people with AIDS, are prone to severe complications from HSV infections. HSV infection has also been associated with cognitive deficits of bipolar disorder,[15] and Alzheimer's disease, although this is often dependent on the genetics of the infected person.
In all cases, HSV is never removed from the body by the immune system. Following a primary infection, the virus enters the nerves at the site of primary infection, migrates to the cell body of the neuron, and becomes latent in the ganglion.[16] As a result of primary infection, the body produces antibodies to the particular type of HSV involved, which can help reduce the odds of subsequent infection of that type at a different site. In HSV-1-infected individuals, seroconversion after an oral infection helps prevent additional HSV-1 infections such as whitlow, genital herpes, and herpes of the eye. Prior HSV-1 seroconversion seems to reduce the symptoms of a later HSV-2 infection, although HSV-2 can still be contracted.
Many people infected with HSV-2 display no physical symptoms—individuals with no symptoms are described as asymptomatic or as having subclinical herpes.[17] However, infection with herpes can be fatal.[18]
Types of herpes
| Condition | Description | Illustration |
|---|---|---|
| Herpetic gingivostomatitis | Herpetic gingivostomatitis is often the initial presentation during the first herpes infection. It is of greater severity than herpes labialis, which is often the subsequent presentation. |  |
| Herpes labialis | Commonly referred to as cold sores or fever blisters, herpes labialis is the most common presentation of recurrent HSV-1 infection following the re-emergence of the virus from the trigeminal nerve. |  |
| Herpes genitalis | When symptomatic, the typical manifestation of a primary HSV-1 or HSV-2 genital infection is clusters of inflamed papules and vesicles on the outer surface of the genitals resembling cold sores. |  |
| Herpetic whitlow and herpes gladiatorum | Herpes whitlow is a painful infection that typically affects the fingers or thumbs. On occasion, infection occurs on the toes or the nail cuticle. Individuals who participate in contact sports such as wrestling, rugby, and football (soccer), sometimes acquire a condition caused by HSV-1 known as herpes gladiatorum, scrumpox, wrestler's herpes, or mat herpes, which presents as skin ulceration on the face, ears, and neck. Symptoms include fever, headache, sore throat, and swollen glands. It occasionally affects the eyes or eyelids. |  |
| Herpesviral encephalitis and herpesviral meningitis | Herpes simplex encephalitis (HSE) is a rare life-threatening condition that is thought to be caused by the transmission of HSV-1 either from the nasal cavity to the brain's temporal lobe or from a peripheral site on the face, along the trigeminal nerve axon, to the brainstem.[19][20][21][22] Despite its low incidence, HSE is the most common sporadic fatal encephalitis worldwide. HSV-2 is the most common cause of Mollaret's meningitis, a type of recurrent viral meningitis. |  |
| Herpes esophagitis | Symptoms may include painful swallowing (odynophagia) and difficulty swallowing (dysphagia). It is often associated with impaired immune function (e.g. HIV/AIDS, immunosuppression in solid organ transplants). |  |
Other
Neonatal herpes simplex is an HSV infection in an infant. It is a rare but serious condition, usually caused by vertical transmission of HSV-1 or -2 from mother to newborn. During immunodeficiency, herpes simplex can cause unusual lesions in the skin. One of the most striking is the appearance of clean linear erosions in skin creases, with the appearance of a knife cut.[23] Herpetic sycosis is a recurrent or initial herpes simplex infection affecting primarily the hair follicles.[24]: 369 Eczema herpeticum is an infection with herpesvirus in patients with chronic atopic dermatitis may result in spread of herpes simplex throughout the eczematous areas.[24]: 373
Herpetic keratoconjunctivitis, a primary infection, typically presents as swelling of the conjunctiva and eyelids (blepharoconjunctivitis), accompanied by small white itchy lesions on the surface of the cornea.
Herpetic sycosis is a recurrent or initial herpes simplex infection affecting primarily the hair follicle.[24]: 369 [25]
Bell's palsy
Although the exact cause of Bell's palsy—a type of facial paralysis—is unknown, it may be related to the reactivation of HSV-1.[26] This theory has been contested, however, since HSV is detected in large numbers of individuals having never experienced facial paralysis, and higher levels of antibodies for HSV are not found in HSV-infected individuals with Bell's palsy compared to those without.[27] Antivirals may improve the condition slightly when used together with corticosteroids in those with severe disease.[28]
Alzheimer's disease
HSV-1 has been proposed as a possible cause of Alzheimer's disease.[29][30] In the presence of a certain gene variation (APOE-epsilon4 allele carriers), HSV-1 appears to be particularly damaging to the nervous system and increases one's risk of developing Alzheimer's disease. The virus interacts with the components and receptors of lipoproteins, which may lead to its development.[31][32]
Pathophysiology
| HSV-2 genital | 15–25% of days |
| HSV-1 oral | 6–33% of days |
| HSV-1 genital | 5% of days |
| HSV-2 oral | 1% of days |
Herpes is contracted through direct contact with an active lesion or body fluid of an infected person.[34] Herpes transmission occurs between discordant partners; a person with a history of infection (HSV seropositive) can pass the virus to an HSV seronegative person. Herpes simplex virus 2 is typically contracted through direct skin-to-skin contact with an infected individual, but can also be contracted by exposure to infected saliva, semen, vaginal fluid, or the fluid from herpetic blisters.[35] To infect a new individual, HSV travels through tiny breaks in the skin or mucous membranes in the mouth or genital areas. Even microscopic abrasions on mucous membranes are sufficient to allow viral entry.
HSV asymptomatic shedding occurs at some time in most individuals infected with herpes. It can occur more than a week before or after a symptomatic recurrence in 50% of cases.[36] Virus enters into susceptible cells by entry receptors[37] such as nectin-1, HVEM and 3-O sulfated heparan sulfate.[38] Infected people who show no visible symptoms may still shed and transmit viruses through their skin; asymptomatic shedding may represent the most common form of HSV-2 transmission.[36] Asymptomatic shedding is more frequent within the first 12 months of acquiring HSV. Concurrent infection with HIV increases the frequency and duration of asymptomatic shedding.[39] Some individuals may have much lower patterns of shedding, but evidence supporting this is not fully verified; no significant differences are seen in the frequency of asymptomatic shedding when comparing persons with one to 12 annual recurrences to those with no recurrences.[36]
Antibodies that develop following an initial infection with a type of HSV can reduce the odds of reinfection with the same virus type.[40] In a monogamous couple, a seronegative female runs a greater than 30% per year risk of contracting an HSV infection from a seropositive male partner.[41] If an oral HSV-1 infection is contracted first, seroconversion will have occurred after 6 weeks to provide protective antibodies against a future genital HSV-1 infection.[40] Herpes simplex is a double-stranded DNA virus.[42]
Diagnosis
Classification
Herpes simplex virus is divided into two types.[4] However, each may cause infections in all areas.[4]
- HSV-1 causes primarily mouth, throat, face, eye, and central nervous system infections.[4]
- HSV-2 causes primarily anogenital infections.[4]
Examination
Primary orofacial herpes is readily identified by examination of persons with no previous history of lesions and contact with an individual with known HSV infection. The appearance and distribution of sores is typically presents as multiple, round, superficial oral ulcers, accompanied by acute gingivitis.[43] Adults with atypical presentation are more difficult to diagnose. Prodromal symptoms that occur before the appearance of herpetic lesions help differentiate HSV symptoms from the similar symptoms of other disorders, such as allergic stomatitis. When lesions do not appear inside the mouth, primary orofacial herpes is sometimes mistaken for impetigo, a bacterial infection. Common mouth ulcers (aphthous ulcer) also resemble intraoral herpes, but do not present a vesicular stage.[43]
Genital herpes can be more difficult to diagnose than oral herpes, since most people have none of the classical symptoms.[43] Further confusing diagnosis, several other conditions resemble genital herpes, including fungal infection, lichen planus, atopic dermatitis, and urethritis.[43]
Laboratory testing
Laboratory testing is often used to confirm a diagnosis of genital herpes. Laboratory tests include culture of the virus, direct fluorescent antibody (DFA) studies to detect virus, skin biopsy, and polymerase chain reaction to test for presence of viral DNA. Although these procedures produce highly sensitive and specific diagnoses, their high costs and time constraints discourage their regular use in clinical practice.[43]
Until the 1980s serological tests for antibodies to HSV were rarely useful to diagnosis and not routinely used in clinical practice.[43] The older IgM serologic assay could not differentiate between antibodies generated in response to HSV-1 or HSV-2 infection. However, a glycoprotein G-specific (IgG) HSV test introduced in the 1980s is more than 98% specific at discriminating HSV-1 from HSV-2.[44]
Differential diagnosis
It should not be confused with conditions caused by other viruses in the herpesviridae family such as herpes zoster (also known as shingles), which is caused by varicella zoster virus. The differential diagnosis includes hand, foot and mouth disease due to similar lesions on the skin. Lymphangioma circumscriptum and dermatitis herpetiformis may also have a similar appearance.
Prevention
As with almost all sexually transmitted infections, women are more susceptible to acquiring genital HSV-2 than men.[45] On an annual basis, without the use of antivirals or condoms, the transmission risk of HSV-2 from infected male to female is about 8–11%.[41][46] This is believed to be due to the increased exposure of mucosal tissue to potential infection sites. Transmission risk from infected female to male is around 4–5% annually.[46] Suppressive antiviral therapy reduces these risks by 50%.[47] Antivirals also help prevent the development of symptomatic HSV in infection scenarios, meaning the infected partner will be seropositive but symptom-free by about 50%. Condom use also reduces the transmission risk significantly.[48][49] Condom use is much more effective at preventing male-to-female transmission than vice versa.[48] Previous HSV-1 infection may reduce the risk for acquisition of HSV-2 infection among women by a factor of three, although the one study that states this has a small sample size of 14 transmissions out of 214 couples.[50]
However, asymptomatic carriers of the HSV-2 virus are still contagious. In many infections, the first symptom people will have of their own infections is the horizontal transmission to a sexual partner or the vertical transmission of neonatal herpes to a newborn at term. Since most asymptomatic individuals are unaware of their infection, they are considered at high risk for spreading HSV.[51]
In October 2011, the anti-HIV drug tenofovir, when used topically in a microbicidal vaginal gel, was reported to reduce herpes virus sexual transmission by 51%.[52]
Barrier methods
Condoms offer moderate protection against HSV-2 in both men and women, with consistent condom users having a 30%-lower risk of HSV-2 acquisition compared with those who never use condoms.[53] A female condom can provide greater protection than the male condom, as it covers the labia.[54] The virus cannot pass through a synthetic condom, but a male condom's effectiveness is limited[55] because herpes ulcers may appear on areas not covered by it. Neither type of condom prevents contact with the scrotum, anus, buttocks, or upper thighs, areas that may come in contact with ulcers or genital secretions during sexual activity. Protection against herpes simplex depends on the site of the ulcer; therefore, if ulcers appear on areas not covered by condoms, abstaining from sexual activity until the ulcers are fully healed is one way to limit risk of transmission.[56] The risk is not eliminated, however, as viral shedding capable of transmitting infection may still occur while the infected partner is asymptomatic.[57] The use of condoms or dental dams also limits the transmission of herpes from the genitals of one partner to the mouth of the other (or vice versa) during oral sex. When one partner has a herpes simplex infection and the other does not, the use of antiviral medication, such as valaciclovir, in conjunction with a condom, further decreases the chances of transmission to the uninfected partner.[16] Topical microbicides that contain chemicals that directly inactivate the virus and block viral entry are being investigated.[16]
Antivirals
Antivirals may reduce asymptomatic shedding; asymptomatic genital HSV-2 viral shedding is believed to occur on 20% of days per year in patients not undergoing antiviral treatment, versus 10% of days while on antiviral therapy.[36]
Pregnancy
The risk of transmission from mother to baby is highest if the mother becomes infected around the time of delivery (30% to 60%),[58][59] since insufficient time will have occurred for the generation and transfer of protective maternal antibodies before the birth of the child. In contrast, the risk falls to 3% if the infection is recurrent,[60] and is 1–3% if the woman is seropositive for both HSV-1 and HSV-2,[60][61] and is less than 1% if no lesions are visible.[60] Women seropositive for only one type of HSV are only half as likely to transmit HSV as infected seronegative mothers. To prevent neonatal infections, seronegative women are recommended to avoid unprotected oral-genital contact with an HSV-1-seropositive partner and conventional sex with a partner having a genital infection during the last trimester of pregnancy. Mothers infected with HSV are advised to avoid procedures that would cause trauma to the infant during birth (e.g. fetal scalp electrodes, forceps, and vacuum extractors) and, should lesions be present, to elect caesarean section to reduce exposure of the child to infected secretions in the birth canal.[16] The use of antiviral treatments, such as aciclovir, given from the 36th week of pregnancy, limits HSV recurrence and shedding during childbirth, thereby reducing the need for caesarean section.[16]
Aciclovir is the recommended antiviral for herpes suppressive therapy during the last months of pregnancy. The use of valaciclovir and famciclovir, while potentially improving compliance, have less-well-determined safety in pregnancy.
Management
No method eradicates herpes virus from the body, but antiviral medications can reduce the frequency, duration, and severity of outbreaks. Analgesics such as ibuprofen and paracetamol (acetaminophen) can reduce pain and fever. Topical anesthetic treatments such as prilocaine, lidocaine, benzocaine, or tetracaine can also relieve itching and pain.[62][63][64]
Antiviral

Several antiviral drugs are effective for treating herpes, including aciclovir (acyclovir), valaciclovir, famciclovir, and penciclovir. Aciclovir was the first discovered and is now available in generic.[65] Valaciclovir is also available as a generic[66] and is slightly more effective than aciclovir for reducing lesion healing time.[67]
Evidence supports the use of aciclovir and valaciclovir in the treatment of herpes labialis[68] as well as herpes infections in people with cancer.[69] The evidence to support the use of aciclovir in primary herpetic gingivostomatitis is weaker.[70]
Topical
A number of topical antivirals are effective for herpes labialis, including aciclovir, penciclovir, and docosanol.[68][71]
Alternative medicine
Evidence is insufficient to support use of many of these compounds, including echinacea, eleuthero, L-lysine, zinc, monolaurin bee products, and aloe vera.[72] While a number of small studies show possible benefit from monolaurin, L-lysine, aspirin, lemon balm, topical zinc, or licorice root cream in treatment, these preliminary studies have not been confirmed by higher-quality randomized controlled studies.[73]
Prognosis
Following active infection, herpes viruses establish a latent infection in sensory and autonomic ganglia of the nervous system. The double-stranded DNA of the virus is incorporated into the cell physiology by infection of the nucleus of a nerve's cell body. HSV latency is static; no virus is produced; and is controlled by a number of viral genes, including latency-associated transcript.[74]
Many HSV-infected people experience recurrence within the first year of infection.[16] Prodrome precedes development of lesions. Prodromal symptoms include tingling (paresthesia), itching, and pain where lumbosacral nerves innervate the skin. Prodrome may occur as long as several days or as short as a few hours before lesions develop. Beginning antiviral treatment when prodrome is experienced can reduce the appearance and duration of lesions in some individuals. During recurrence, fewer lesions are likely to develop and are less painful and heal faster (within 5–10 days without antiviral treatment) than those occurring during the primary infection.[16] Subsequent outbreaks tend to be periodic or episodic, occurring on average four or five times a year when not using antiviral therapy.
The causes of reactivation are uncertain, but several potential triggers have been documented. A 2009 study showed the protein VP16 plays a key role in reactivation of the dormant virus.[75] Changes in the immune system during menstruation may play a role in HSV-1 reactivation.[76][77] Concurrent infections, such as viral upper respiratory tract infection or other febrile diseases, can cause outbreaks. Reactivation due to other infections is the likely source of the historic terms 'cold sore' and 'fever blister'.
Other identified triggers include local injury to the face, lips, eyes, or mouth; trauma; surgery; radiotherapy; and exposure to wind, ultraviolet light, or sunlight.[78][79][80][81][82]
The frequency and severity of recurrent outbreaks vary greatly between people. Some individuals' outbreaks can be quite debilitating, with large, painful lesions persisting for several weeks, while others experience only minor itching or burning for a few days. Some evidence indicates genetics play a role in the frequency of cold sore outbreaks. An area of human chromosome 21 that includes six genes has been linked to frequent oral herpes outbreaks. An immunity to the virus is built over time. Most infected individuals experience fewer outbreaks and outbreak symptoms often become less severe. After several years, some people become perpetually asymptomatic and no longer experience outbreaks, though they may still be contagious to others. Immunocompromised individuals may experience longer, more frequent, and more severe episodes. Antiviral medication has been proven to shorten the frequency and duration of outbreaks.[83] Outbreaks may occur at the original site of the infection or in proximity to nerve endings that reach out from the infected ganglia. In the case of a genital infection, sores can appear at the original site of infection or near the base of the spine, the buttocks, or the back of the thighs. HSV-2-infected individuals are at higher risk for acquiring HIV when practicing unprotected sex with HIV-positive persons, in particular during an outbreak with active lesions.[84]
Epidemiology
Worldwide rates of either HSV-1 and/or HSV-2 are between 60 and 95% in adults.[4] HSV-1 is more common than HSV-2, with rates of both increasing as people age.[4] HSV-1 rates are between 70% and 80% in populations of low socioeconomic status and 40% to 60% in populations of improved socioeconomic status.[4] An estimated 536 million people or 16% of the population worldwide were infected with HSV-2 as of 2003 with greater rates among women and in those in the developing world.[12] Rates of infection are determined by the presence of antibodies against either viral species.[85]
In the US, 58% of the population is infected with HSV-1[86] and 16% are infected with HSV-2. Among those HSV-2-seropositive, only 19% were aware they were infected.[87] During 2005–2008, the prevalence of HSV-2 was 39% in black people and 21% in women.[88]
The annual incidence in Canada of genital herpes due to HSV-1 and HSV-2 infection is not known (for a review of HSV-1/HSV-2 prevalence and incidence studies worldwide, see Smith and Robinson 2002). As many as one in seven Canadians aged 14 to 59 may be infected with herpes simplex type 2 virus[89] and more than 90 per cent of them may be unaware of their status, a new study suggests.[90] In the United States, it is estimated that about 1,640,000 HSV-2 seroconversions occur yearly (730,000 men and 910,000 women, or 8.4 per 1,000 persons).[91]
In British Columbia in 1999, the seroprevalence of HSV-2 antibody in leftover serum submitted for antenatal testing revealed a prevalence of 17%, ranging from 7% in women 15–19 years old to 28% in those 40–44 years.[92]
In Norway, a study published in 2000 found that up to 70–90% of genital initial infections were due to HSV-1.[93]
In Nova Scotia, 58% of 1,790 HSV isolates from genital lesion cultures in women were HSV-1; in men, 37% of 468 isolates were HSV-1.[94]
History
Herpes originated and evolved in Africa and could be the result of a cross-species transmission event from gibbons, orangutans, or gorillas.[95]
Herpes has been known for at least 2,000 years. Emperor Tiberius is said to have banned kissing in Rome for a time due to so many people having cold sores. In the 16th century Romeo and Juliet, blisters "o'er ladies' lips" are mentioned. In the 18th century, it was so common among prostitutes that it was called "a vocational disease of women".[96] The term 'herpes simplex' appeared in Richard Boulton's A System of Rational and Practical Chirurgery in 1713, where the terms 'herpes miliaris' and 'herpes exedens' also appeared. Herpes was not found to be a virus until the 1940s.[96]
Herpes antiviral therapy began in the early 1960s with the experimental use of medications that interfered with viral replication called deoxyribonucleic acid (DNA) inhibitors. The original use was against normally fatal or debilitating illnesses such as adult encephalitis,[97] keratitis,[98] in immunocompromised (transplant) patients,[99] or disseminated herpes zoster (also known as disseminated shingles).[100] The original compounds used were 5-iodo-2'-deoxyuridine, AKA idoxuridine, IUdR, or(IDU) and 1-β-D-arabinofuranosylcytosine or ara-C,[101] later marketed under the name cytosar or cytarabine. The usage expanded to include topical treatment of herpes simplex,[102] zoster, and varicella.[103] Some trials combined different antivirals with differing results.[97] The introduction of 9-β-D-arabinofuranosyladenine, (ara-A or vidarabine), considerably less toxic than ara-C, in the mid-1970s, heralded the way for the beginning of regular neonatal antiviral treatment. Vidarabine was the first systemically administered antiviral medication with activity against HSV for which therapeutic efficacy outweighed toxicity for the management of life-threatening HSV disease. Intravenous vidarabine was licensed for use by the U.S. Food and Drug Administration in 1977. Other experimental antivirals of that period included: heparin,[104] trifluorothymidine (TFT),[105] Ribivarin,[106] interferon,[107] Virazole,[108] and 5-methoxymethyl-2'-deoxyuridine (MMUdR).[109] The introduction of 9-(2-hydroxyethoxymethyl)guanine, AKA aciclovir, in the late 1970s[110] raised antiviral treatment another notch and led to vidarabine vs. aciclovir trials in the late 1980s.[111] The lower toxicity and ease of administration over vidarabine has led to aciclovir becoming the drug of choice for herpes treatment after it was licensed by the FDA in 1998.[112] Another advantage in the treatment of neonatal herpes included greater reductions in mortality and morbidity with increased dosages, which did not occur when compared with increased dosages of vidarabine.[112] However, aciclovir seems to inhibit antibody response, and newborns on aciclovir antiviral treatment experienced a slower rise in antibody titer than those on vidarabine.[112]
Society and culture
Some people experience negative feelings related to the condition following diagnosis, in particular, if they have acquired the genital form of the disease. Feelings can include depression, fear of rejection, feelings of isolation, fear of being found out, and self-destructive feelings.[113] Herpes support groups have been formed in the United States and the United Kingdom, providing information about herpes and running message forums and dating websites for affected people. People with the herpes virus are often hesitant to divulge to other people, including friends and family, that they are infected. This is especially true of new or potential sexual partners whom they consider casual.[114]
In a 2007 study, 1,900 people (25% of which had herpes) ranked genital herpes second for social stigma, out of all sexually transmitted diseases (HIV took the top spot for STD stigma).[115][116][117]
Support groups
United States
A source of support is the National Herpes Resource Center which arose from the work of the American Sexual Health Association (ASHA).[118] The ASHA was created in 1914 in response to the increase in sexually transmitted diseases that had spread during World War I.[119] During the 1970s, there was an increase in sexually transmitted diseases. One of the diseases that increased dramatically was genital herpes. In response, ASHA created the National Herpes Resource Center in 1979. The Herpes Resource Center (HRC) was designed to meet the growing need for education and awareness about the virus. One of the projects of the HRC was to create a network of local support (HELP) groups. The goal of these HELP groups was to provide a safe, confidential environment where participants can get accurate information and share experiences, fears, and feelings with others who are concerned about herpes.[120][121]
UK
In the UK, the Herpes Association (now the Herpes Viruses Association) was started in 1982, becoming a registered charity with a Department of Health grant in 1985. The charity started as a string of local group meetings before acquiring an office and a national spread.[122]
Research
Research has gone into vaccines for both prevention and treatment of herpes infections.
As of October 2022, the U.S. FDA have not approved a vaccine for herpes.[123] However, there are herpes vaccines currently in clinical trials, such as Moderna mRNA-1608.[124] Unsuccessful clinical trials have been conducted for some glycoprotein subunit vaccines.[citation needed] As of 2017, the future pipeline includes several promising replication-incompetent vaccine proposals while two replication-competent (live-attenuated) HSV vaccine are undergoing human testing.[citation needed]
A genomic study of the herpes simplex type 1 virus confirmed the human migration pattern theory known as the out-of-Africa hypothesis.[125]
References
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z "Genital Herpes – CDC Fact Sheet". cdc.gov. December 8, 2014. Archived from the original on 31 December 2014. Retrieved 31 December 2014.
- ^ a b c d e f g h i j Balasubramaniam, R; Kuperstein, AS; Stoopler, ET (April 2014). "Update on oral herpes virus infections". Dental Clinics of North America. 58 (2): 265–80. doi:10.1016/j.cden.2013.12.001. PMID 24655522.
- ^ a b Elad S, Zadik Y, Hewson I, et al. (August 2010). "A systematic review of viral infections associated with oral involvement in cancer patients: a spotlight on Herpesviridea". Support Care Cancer. 18 (8): 993–1006. doi:10.1007/s00520-010-0900-3. PMID 20544224. S2CID 2969472.
- ^ a b c d e f g h i j k Chayavichitsilp P, Buckwalter JV, Krakowski AC, Friedlander SF (April 2009). "Herpes simplex". Pediatr Rev. 30 (4): 119–29, quiz 130. doi:10.1542/pir.30-4-119. PMID 19339385. S2CID 34735917.
- ^ "Herpes simplex virus". World Health Organization. Retrieved 21 November 2022.
- ^ Mosby (2013). Mosby's Medical Dictionary (9 ed.). Elsevier Health Sciences. pp. 836–37. ISBN 9780323112581. Archived from the original on 2017-09-06.
- ^ Wu, IB; Schwartz, RA (March 2007). "Herpetic whitlow". Cutis. 79 (3): 193–06. PMID 17674583.
- ^ Rowe, AM; St Leger, AJ; Jeon, S; Dhaliwal, DK; Knickelbein, JE; Hendricks, RL (January 2013). "Herpes keratitis". Progress in Retinal and Eye Research. 32: 88–101. doi:10.1016/j.preteyeres.2012.08.002. PMC 3529813. PMID 22944008.
- ^ Steiner, I; Benninger, F (December 2013). "Update on herpes virus infections of the nervous system". Current Neurology and Neuroscience Reports. 13 (12): 414. doi:10.1007/s11910-013-0414-8. PMID 24142852. S2CID 22139709.
- ^ Stephenson-Famy, A; Gardella, C (December 2014). "Herpes Simplex Virus Infection During Pregnancy". Obstetrics and Gynecology Clinics of North America. 41 (4): 601–14. doi:10.1016/j.ogc.2014.08.006. PMID 25454993.
- ^ Hoyle, Alice; McGeeney, Ester (2019). Great Relationships and Sex Education. Taylor and Francis. ISBN 978-1-35118-825-8. Retrieved July 11, 2023.
- ^ a b Looker, KJ; Garnett, GP; Schmid, GP (October 2008). "An estimate of the global prevalence and incidence of herpes simplex virus type 2 infection". Bulletin of the World Health Organization. 86 (10): 805–12, A. doi:10.2471/blt.07.046128. PMC 2649511. PMID 18949218.
- ^ Beswick, TSL (1962). "The Origin and the Use of the Word Herpes". Med Hist. 6 (3): 214–232. doi:10.1017/S002572730002737X. PMC 1034725. PMID 13868599.
- ^ Reese, Vail. "Countering Creeping Confusion: A Proposal to Re-Name Herpes Virus TAXONOMY". Online Journal of Community and Person-Centered Dermatology. Dr. David Elpern. Archived from the original on 23 September 2018. Retrieved 22 September 2018.
- ^ Dickerson FB, Boronow JJ, Stallings C, et al. (March 2004). "Infection with herpes simplex virus type 1 is associated with cognitive deficits in bipolar disorder". Biol. Psychiatry. 55 (6): 588–93. doi:10.1016/j.biopsych.2003.10.008. PMID 15013827. S2CID 25338399.
- ^ a b c d e f g Gupta R, Warren T, Wald A (December 2007). "Genital herpes". Lancet. 370 (9605): 2127–37. doi:10.1016/S0140-6736(07)61908-4. PMID 18156035. S2CID 40916450.
- ^ Handsfield HH (2000). "Public Health Strategies to Prevent Genital Herpes: Where Do We Stand?". Curr Infect Dis Rep. 2 (1): 25–30. doi:10.1007/s11908-000-0084-y. PMID 11095834. S2CID 41426466.
- ^ Marsh, Sarah (30 December 2021). "Inquests to be held into deaths of new mothers who died from herpes". The Guardian.
- ^ Herpes Encephalitis at eMedicine
- ^ van Riel, Debby; Verdijk, Rob; Kuiken, Thijs (January 2015). "The olfactory nerve: a shortcut for influenza and other viral diseases into the central nervous system". The Journal of Pathology. 235 (2): 277–287. doi:10.1002/path.4461. ISSN 1096-9896. PMID 25294743. S2CID 22929529.
- ^ Esiri, M. M. (May 1982). "Herpes simplex encephalitis. An immunohistological study of the distribution of viral antigen within the brain". Journal of the Neurological Sciences. 54 (2): 209–226. doi:10.1016/0022-510X(82)90183-6. ISSN 0022-510X. PMID 6284882. S2CID 20325355.
- ^ Whitley, R. J.; Soong, S. J.; Linneman, C.; Liu, C.; Pazin, G.; Alford, C. A. (1982-01-15). "Herpes simplex encephalitis. Clinical Assessment". JAMA. 247 (3): 317–320. doi:10.1001/jama.1982.03320280037026. ISSN 0098-7484. PMID 6275134.
- ^ Jocelyn A. Lieb; Stacey Brisman; Sara Herman; Jennifer MacGregor; Marc E. Grossman (2008). "Linear erosive Herpes Simplex Virus infection in immunocompromised patients: the "Knife-Cut Sign"". Clin Infect Dis. 47 (11): 1440–41. doi:10.1086/592976. PMID 18937574.
- ^ a b c James, William D.; Berger, Timothy G. (2006). Andrews' Diseases of the Skin: clinical Dermatology. Saunders Elsevier. ISBN 978-0-7216-2921-6.
- ^ Rapini, Ronald P.; Bolognia, Jean L.; Jorizzo, Joseph L. (2007). Dermatology: 2-Volume Set. St. Louis: Mosby. ISBN 978-1-4160-2999-1.
- ^ Tankéré F, Bernat I (September 2009). "[Bell's palsy: from viral aetiology to diagnostic reality]". Rev Méd Interne (in French). 30 (9): 769–75. doi:10.1016/j.revmed.2008.12.006. PMID 19195745.
- ^ Linder T, Bossart W, Bodmer D (January 2005). "Bell's palsy and Herpes simplex virus: fact or mystery?". Otol. Neurotol. 26 (1): 109–13. doi:10.1097/00129492-200501000-00020. PMID 15699730. S2CID 33873521.
- ^ Gagyor, Ildiko; Madhok, Vishnu B.; Daly, Fergus; Somasundara, Dhruvashree; Sullivan, Michael; Gammie, Fiona; Sullivan, Frank (2015-11-09). "Antiviral treatment for Bell's palsy (idiopathic facial paralysis)" (PDF). The Cochrane Database of Systematic Reviews (11): CD001869. doi:10.1002/14651858.CD001869.pub8. ISSN 1469-493X. PMID 26559436.
- ^ Itzhaki RF, Wozniak MA (May 2008). "Herpes simplex virus type 1 in Alzheimer's disease: the enemy within". J. Alzheimers Dis. 13 (4): 393–405. doi:10.3233/JAD-2008-13405. PMID 18487848.
- ^ Holmes C, Cotterell D (December 2009). "Role of infection in the pathogenesis of Alzheimer's disease: implications for treatment". CNS Drugs. 23 (12): 993–1002. doi:10.2165/11310910-000000000-00000. PMID 19958038. S2CID 25248989.
- ^ Dobson CB, Itzhaki RF (1999). "Herpes simplex virus type 1 and Alzheimer's disease". Neurobiol. Aging. 20 (4): 457–65. doi:10.1016/S0197-4580(99)00055-X. PMID 10604441. S2CID 23633290.
- ^ Pyles RB (2001). "The association of herpes simplex virus and Alzheimer's disease: a potential synthesis of genetic and environmental factors". Herpes. 8 (3): 64–68. PMID 11867022.
- ^ Warren, Terri (2009). The Good News about the Bad News: Herpes: Everything You Need to Know. New Harbinger Publications. p. 28. ISBN 978-1-57224-618-8. Archived from the original on 2016-05-27.
- ^ "AHMF: Preventing Sexual Transmission of Genital herpes". Archived from the original on January 21, 2008. Retrieved 2008-02-24.
- ^ Anita L. Nelson; Jo Ann Woodward (2007-12-14). Sexually Transmitted Diseases: A Practical Guide for Primary Care. Springer Science & Business Media. pp. 50–. ISBN 978-1-59745-040-9.
- ^ a b c d Leone P (2005). "Reducing the risk of transmitting genital herpes: advances in understanding and therapy". Curr Med Res Opin. 21 (10): 1577–82. doi:10.1185/030079905X61901. PMID 16238897. S2CID 26738979.
- ^ Akhtar, Jihan; Shukla, Deepak (December 2009). "Viral entry mechanisms: cellular and viral mediators of herpes simplex virus entry". FEBS Journal. 276 (24): 7228–36. doi:10.1111/j.1742-4658.2009.07402.x. PMC 2801626. PMID 19878306.
- ^ Shukla, Deepak; Liu, Jian; Blaiklock, Peter; Shworak, Nicholas W.; Bai, Xiaomei; Esko, Jeffrey D.; Cohen, Gary H.; Eisenberg, Roselyn; et al. (1999). "A Novel Role for 3-O-Sulfated Heparan Sulfate in Herpes Simplex Virus 1 Entry". Cell. 99 (1): 13–22. doi:10.1016/S0092-8674(00)80058-6. PMID 10520990. S2CID 14139940.
- ^ Kim H, Meier A, Huang M, Kuntz S, Selke S, Celum C, Corey L, Wald A (2006). "Oral herpes simplex virus type 2 reactivation in HIV-positive and -negative men". J Infect Dis. 194 (4): 420–27. doi:10.1086/505879. PMID 16845624.
- ^ a b "Herpes simplex pathophysiology". WikiDoc. Retrieved 2021-03-06.
- ^ a b Mertz, G.J. (1993). "Epidemiology of genital herpes infections". Infect Dis Clin North Am. 7 (4): 825–39. doi:10.1016/S0891-5520(20)30561-4. PMID 8106731.
- ^ Reuven NB, Staire AE, Myers RS, Weller SK (2003). "The herpes simplex virus type 1 alkaline nuclease and single-stranded DNA binding protein mediate strand exchange in vitro". J. Virol. 77 (13): 7425–33. doi:10.1128/jvi.77.13.7425-7433.2003. PMC 164775. PMID 12805441.
- ^ a b c d e f Fatahzadeh M, Schwartz RA (2007). "Human herpes simplex virus infections: epidemiology, pathogenesis, symptomatology, diagnosis, and management". J. Am. Acad. Dermatol. 57 (5): 737–63, quiz 764–6. doi:10.1016/j.jaad.2007.06.027. PMID 17939933.
- ^ Ashley RL, et al. (1988). "Comparison of Western blot (immunoblot) and glycoprotein G-specific immunodot enzyme assay for detecting antibodies to herpes simplex virus types 1 and 2 in human sera". J. Clin. Microbiol. 26 (4): 662–67. doi:10.1128/JCM.26.4.662-667.1988. PMC 266403. PMID 2835389.
- ^ Carla K. Johnson (August 23, 2006). "Percentage of people with herpes drops". Associated Press. Archived from the original on 2012-03-18. Retrieved 2011-04-12.
- ^ a b Kulhanjian J A, Soroush V, Au DS, et al. (April 2, 1992). "Identification of women at unsuspected risk of primary infection with herpes simplex virus type 2 during pregnancy". N. Engl. J. Med. 326 (14): 916–20. doi:10.1056/NEJM199204023261403. PMID 1311799.
- ^ Corey L, Wald A, Patel R, et al. (January 2004). "Once-daily valacyclovir to reduce the risk of transmission of genital herpes". N Engl J Med. 350 (1): 11–20. doi:10.1056/NEJMoa035144. PMID 14702423. S2CID 21573428.
- ^ a b Wald A, Langenberg AG, Link K, Izu AE, Ashley R, Warren T, Tyring S, Douglas JM Jr, Corey L (2001). "Effect of condoms on reducing the transmission of herpes simplex virus type 2 from men to women". JAMA. 285 (24): 3100–06. doi:10.1001/jama.285.24.3100. PMID 11427138.
- ^ Wald A, Langenberg AG, Krantz E, et al. (November 2005). "The relationship between condom use and herpes simplex virus acquisition". Annals of Internal Medicine. 143 (10): 707–13. doi:10.7326/0003-4819-143-10-200511150-00007. PMID 16287791. S2CID 37342783.
- ^ Mertz, GJ; Benedetti J; Ashley R; Selke SA; Corey L. (1 February 1992). "Risk factors for the sexual transmission of genital herpes". Annals of Internal Medicine. 116 (3): 197–202. doi:10.7326/0003-4819-116-3-197. PMID 1309413. S2CID 40143915.
- ^ "Genital Herpes – CDC Fact Sheet". Center for Disease Control and Prevention. Archived from the original on 2014-01-30. Retrieved 2014-01-30.
- ^ McNeil DG. Topical Tenofovir, a Microbicide Effective against HIV, Inhibits Herpes Simplex Virus-2 Replication Archived 2017-04-09 at the Wayback Machine. NY Times. Research article: Andrei G; Lisco A; Vanpouille C; et al. (October 2011). "Topical Tenofovir, a Microbicide Effective against HIV, Inhibits Herpes Simplex Virus-2 Replication". Cell Host & Microbe. 10 (4): 379–89. doi:10.1016/j.chom.2011.08.015. PMC 3201796. PMID 22018238.
- ^ Martin ET, Krantz E, Gottlieb SL, Magaret AS, Langenberg A, Stanberry L, Kamb M, Wald A (July 2009). "A pooled analysis of the effect of condoms in preventing HSV-2 acquisition". Archives of Internal Medicine. 169 (13): 1233–40. doi:10.1001/archinternmed.2009.177. PMC 2860381. PMID 19597073.
- ^ "Putting Herpes in Perspective". UBM Medica. Archived from the original on 25 February 2021. Retrieved 20 July 2011.
- ^ "Condom Effectiveness – Male Latex Condoms and Sexually Transmitted Diseases". Center for Disease Control and Prevention. Archived from the original on 2011-10-02. Retrieved 2011-10-01.
- ^ "STD Facts – Genital Herpes". Center for Disease Control and Prevention. Archived from the original on 2011-10-01. Retrieved 2011-10-01.
- ^ Koelle, D.M.; Wald, A. (April 2000). "Herpes simplex virus: The importance of asymptomatic shedding". J. Antimicrob. Chemother. 45 (Suppl T3): 1–8. doi:10.1093/jac/45.suppl_4.1. PMID 10855766.
- ^ Brown ZA, Selke S, Zeh J, et al. (1997). "The acquisition of herpes simplex virus during pregnancy". N Engl J Med. 337 (8): 509–15. doi:10.1056/NEJM199708213370801. PMID 9262493.
- ^ Brown ZA, Wald A, Morrow RA, Selke S, Zeh J, Corey L (2003). "Effect of serologic status and cesarean delivery on transmission rates of herpes simplex virus from mother to infant". JAMA. 289 (2): 203–09. doi:10.1001/jama.289.2.203. PMID 12517231.
- ^ a b c Brown ZA, Benedetti J, Ashley R, et al. (May 1991). "Neonatal herpes simplex virus infection in relation to asymptomatic maternal infection at the time of labor". N. Engl. J. Med. 324 (18): 1247–52. doi:10.1056/NEJM199105023241804. PMID 1849612.
- ^ Whitley RJ, Kimberlin DW, Roizman B (1998). "Herpes simplex viruses". Clin Infect Dis. 26 (3): 541–53. doi:10.1086/514600. PMID 9524821.
- ^ O'Mahony C, Timms MS, Ramsden RT (December 1988). "Local anesthetic creams". BMJ. 297 (6661): 1468. doi:10.1136/bmj.297.6661.1468-a. PMC 1835116. PMID 3147021.
- ^ Kaminester LH, Pariser RJ, Pariser DM, et al. (December 1999). "A double-blind, placebo-controlled study of topical tetracaine in the treatment of herpes labialis". J. Am. Acad. Dermatol. 41 (6): 996–1001. doi:10.1016/S0190-9622(99)70260-4. PMID 10570387.
- ^ Leung DT, Sacks SL (October 2003). "Current treatment options to prevent perinatal transmission of herpes simplex virus". Expert Opin Pharmacother. 4 (10): 1809–19. doi:10.1517/14656566.4.10.1809. PMID 14521490. S2CID 33261337.
- ^ Robert L. LaFemina (2009). Antiviral research : strategies in antiviral drug discovery. Washington, DC: ASM Press. p. 1. ISBN 978-1-55581-439-7. Archived from the original on 2016-05-02.
- ^ Agrawal, Caroline A. Hastings, Joseph Torkildson, Anurag Kishor (2012-04-30). Handbook of pediatric hematology and oncology : Children's Hospital & Research Center Oakland (2nd ed.). Chichester, West Sussex: Wiley-Blackwell. p. 360. ISBN 978-0-470-67088-0. Archived from the original on 2016-04-30.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ Chen, Fangman; Xu, Hao; Liu, Jinli; Cui, Yuan; Luo, Xiaobo; Zhou, Yu; Chen, Qianming; Jiang, Lu (2017). "Efficacy and safety of nucleoside antiviral drugs for treatment of recurrent herpes labialis: a systematic review and meta-analysis". Journal of Oral Pathology & Medicine. 46 (8): 561–568. doi:10.1111/jop.12534. ISSN 0904-2512. PMID 27935123. S2CID 10391761.
- ^ a b Chon T, Nguyen L, Elliott TC (July 2007). "Clinical inquiries. What are the best treatments for herpes labialis?". J Fam Pract. 56 (7): 576–78. PMID 17605952.
- ^ Glenny AM, Fernandez Mauleffinch LM, Pavitt S, Walsh T (January 2009). "Interventions for the prevention and treatment of herpes simplex virus in patients being treated for cancer". The Cochrane Database of Systematic Reviews (1): CD006706. doi:10.1002/14651858.CD006706.pub2. PMID 19160295.
- ^ Nasser M, Fedorowicz Z, Khoshnevisan MH, Shahiri Tabarestani M (October 2008). Nasser M (ed.). "Acyclovir for treating primary herpetic gingivostomatitis". The Cochrane Database of Systematic Reviews (4): CD006700. doi:10.1002/14651858.CD006700.pub2. PMID 18843726. (Retracted, see doi:10.1002/14651858.CD006700.pub3, PMID 26784280, Retraction Watch)
- ^ Treister NS, Woo SB (April 2010). "Topical n-docosanol for management of recurrent herpes labialis". Expert Opin Pharmacother. 11 (5): 853–60. doi:10.1517/14656561003691847. PMID 20210688. S2CID 26237384.
- ^ Perfect MM, Bourne N, Ebel C, Rosenthal SL (October 2005). "Use of complementary and alternative medicine for the treatment of genital herpes". Herpes. 12 (2): 38–41. PMID 16209859.
- ^ Beauman, JG (Oct 15, 2005). "Genital herpes: a review". American Family Physician. 72 (8): 1527–34. PMID 16273819.
- ^ Stumpf MP, Laidlaw Z, Jansen VA (2002). "Herpes viruses hedge their bets". Proc. Natl. Acad. Sci. U.S.A. 99 (23): 15234–37. Bibcode:2002PNAS...9915234S. doi:10.1073/pnas.232546899. PMC 137573. PMID 12409612.
- ^ Thompson, Richard L.; Preston, Chris M.; Sawtell, Nancy M. (2009-03-01). "De novo synthesis of VP16 coordinates the exit from HSV latency in vivo". PLOS Pathogens. 5 (3): e1000352. doi:10.1371/journal.ppat.1000352. ISSN 1553-7374. PMC 2654966. PMID 19325890.
- ^ Myśliwska J, Trzonkowski P, Bryl E, Lukaszuk K, Myśliwski A (2000). "Lower interleukin-2 and higher serum tumor necrosis factor-a levels are associated with perimenstrual, recurrent, facial herpes simplex infection in young women". Eur. Cytokine Netw. 11 (3): 397–406. PMID 11022124.
- ^ Segal AL, Katcher AH, Brightman VJ, Miller MF (1974). "Recurrent herpes labialis, recurrent aphthous ulcers, and the menstrual cycle". J. Dent. Res. 53 (4): 797–803. doi:10.1177/00220345740530040501. PMID 4526372. S2CID 43134857.
- ^ Chambers A, Perry M (2008). "Salivary mediated autoinoculation of herpes simplex virus on the face in the absence of "cold sores," after trauma". J. Oral Maxillofac. Surg. 66 (1): 136–38. doi:10.1016/j.joms.2006.07.019. PMID 18083428.
- ^ Perna JJ, Mannix ML, Rooney JF, Notkins AL, Straus SE (1987). "Reactivation of latent herpes simplex virus infection by ultraviolet light: a human model". J. Am. Acad. Dermatol. 17 (3): 473–78. doi:10.1016/S0190-9622(87)70232-1. PMID 2821086.
- ^ Rooney JF, Straus SE, Mannix ML, et al. (1992). "UV light-induced reactivation of herpes simplex virus type 2 and prevention by acyclovir". J. Infect. Dis. 166 (3): 500–06. doi:10.1093/infdis/166.3.500. PMID 1323616.
- ^ Oakley C, Epstein JB, Sherlock CH (1997). "Reactivation of oral herpes simplex virus: implications for clinical management of herpes simplex virus recurrence during radiotherapy". Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 84 (3): 272–78. doi:10.1016/S1079-2104(97)90342-5. PMID 9377190.
- ^ Ichihashi M, Nagai H, Matsunaga K (2004). "Sunlight is an important causative factor of recurrent herpes simplex". Cutis. 74 (5 Suppl): 14–18. PMID 15603217.
- ^ Martinez V, Caumes E, Chosidow O (2008). "Treatment to prevent recurrent genital herpes". Current Opinion in Infectious Diseases. 21 (1): 42–48. doi:10.1097/QCO.0b013e3282f3d9d3. PMID 18192785. S2CID 25681412.
- ^ Koelle DM, Corey L (2008). "Herpes Simplex: Insights on Pathogenesis and Possible Vaccines". Annu Rev Med. 59: 381–95. doi:10.1146/annurev.med.59.061606.095540. PMID 18186706.
- ^ Smith JS, Robinson NJ (2002). "Age-specific prevalence of infection with herpes simplex virus types 2 and 1: a global review". J. Infect. Dis. 186 (Suppl 1): S3–28. doi:10.1086/343739. PMID 12353183.
- ^ Xu, Fujie; Fujie Xu; Maya R. Sternberg; Benny J. Kottiri; Geraldine M. McQuillan; Francis K. Lee; Andre J. Nahmias; Stuart M. Berman; Lauri E. Markowitz (2006-10-23). "Trends in Herpes Simplex Virus Type 1 and Type 2 Seroprevalence in the United States". JAMA. 296 (8): 964–73. doi:10.1001/jama.296.8.964. PMID 16926356.
- ^ Xu, F; MR Sternberg; SL Gottlieb; SM Berman; LE Markowitz; et al. (23 April 2010). "Seroprevalence of Herpes Simplex Virus Type 2 Among Persons Aged 14–49 Years – United States, 2005–2008". Morbidity and Mortality Weekly Report. 59 (15): 456–59. PMID 20414188. Archived from the original on 25 June 2011. Retrieved 12 April 2011.
- ^ "CDC Study Finds U.S. Herpes Rates Remain High" (PDF). Center for Disease Control and Prevention. 2010-03-09. Archived (PDF) from the original on 2016-03-06. Retrieved 2012-02-19.
- ^ Rotermann, Michelle; Langlois, Kellie A.; Severini, Alberto; Totten, Stephanie (2013-04-01). "Prevalence of Chlamydia trachomatis and herpes simplex virus type 2: Results from the 2009 to 2011 Canadian Health Measures Survey". Health Reports. 24 (4): 10–15. ISSN 1209-1367. PMID 24258059.
- ^ "Herpes virus has infected nearly one in five Canadians over age 35, most unaware they have it: study". National Post. Retrieved 2017-02-20.
- ^ Smith JS, Robinson NJ (2002). "Age-specific prevalence of infection with herpes simplex virus types 2 and 1: a global review". J Infect Dis. 186 (suppl 1): S3–28. doi:10.1086/343739. PMID 12353183.
- ^ Armstrong GL, Schillinger J, Markowitz L, Nahmias AJ, Johnson RE, McQuillan GM, St Louis ME (May 2001). "Incidence of herpes simplex virus type 2 infection in the United States". American Journal of Epidemiology. 153 (9): 912–20. doi:10.1093/aje/153.9.912. PMID 11323323.
- ^ Nilsen A, Myrmel H (2000). "Changing trends in genital herpes simplex virus infection in Bergen, Norway". Acta Obstet Gynecol Scand. 79 (8): 693–96. doi:10.1080/j.1600-0412.2000.079008693.x. PMID 10949236.
- ^ Forward KR, Lee SHS (2003). "Predominance of herpes simplex virus type 1 from patients with genital herpes in Nova Scotia". Can J Infect Dis. 14 (2): 94–96. doi:10.1155/2003/168673. PMC 2094909. PMID 18159431.
- ^ Wertheim, Joel O.; Smith, Martin D.; Smith, Davey M.; Scheffler, Konrad; Kosakovsky Pond, Sergei L. (2014). "Evolutionary Origins of Human Herpes Simplex Viruses 1 and 2". Molecular Biology and Evolution. 31 (9): 2356–2364. doi:10.1093/molbev/msu185. ISSN 1537-1719. PMC 4137711. PMID 24916030.
- ^ a b John Leo (1982-08-02). "The New Scarlet Letter". Time. Archived from the original on 2010-02-02.
- ^ a b Chow AW, Roland A, Fiala M, et al. (March 1973). "Cytosine Arabinoside Therapy for Herpes Simplex Encephalitis – Clinical Experience with Six Patients". Antimicrob. Agents Chemother. 3 (3): 412–17. doi:10.1128/aac.3.3.412. PMC 444424. PMID 4790599.
- ^ Kaufman HE, Howard GM (August 1962). "Therapy of experimental herpes simplex keratitis". Investigative Ophthalmology. 1: 561–4. PMID 14454441.
- ^ Ch'ien LT, Whitley RJ, Alford CA, Galasso GJ (June 1976). "Adenine arabinoside for therapy of herpes zoster in immunosuppressed patients: preliminary results of a collaborative study". J. Infect. Dis. 133 (Suppl): A184–91. doi:10.1093/infdis/133.supplement_2.a184. PMID 180198.
- ^ McKelvey EM, Kwaan HC (November 1969). "Cytosine arabinoside therapy for disseminated herpes zoster in a patient with IgG pyroglobulinemia". Blood. 34 (5): 706–11. doi:10.1182/blood.V34.5.706.706. PMID 5352659.
- ^ Fiala M, Chow A, Guze LB (April 1972). "Susceptibility of Herpesviruses to Cytosine Arabinoside: Standardization of Susceptibility Test Procedure and Relative Resistance of Herpes Simplex Type 2 Strains". Antimicrob. Agents Chemother. 1 (4): 354–57. doi:10.1128/aac.1.4.354. PMC 444221. PMID 4364937.
- ^ Allen LB, Hintz OJ, Wolf SM, et al. (June 1976). "Effect of 9-beta-D-arabinofuranosylhypoxanthine 5'-monophosphate on genital lesions and encephalitis induced by Herpesvirus hominis type 2 in female mice". J. Infect. Dis. 133 (Suppl): A178–83. doi:10.1093/infdis/133.supplement_2.a178. PMID 6598.
- ^ Juel-Jensen BE (March 1970). "Varicella and cytosine arabinoside". Lancet. 1 (7646): 572. doi:10.1016/S0140-6736(70)90815-9. PMID 4190397.
- ^ Nahmias AJ, Kibrick S (May 1964). "Inhibitory Effect of Heparin on Herpes Simplex Virus". J. Bacteriol. 87 (5): 1060–66. doi:10.1128/JB.87.5.1060-1066.1964. PMC 277146. PMID 4289440.
- ^ Allen LB, Sidwell RW (September 1972). "Target-Organ Treatment of Neurotropic Virus Diseases: Efficacy as a Chemotherapy Tool and Comparison of Activity of Adenine Arabinoside, Cytosine Arabinoside, Idoxuridine, and Trifluorothymidine". Antimicrob. Agents Chemother. 2 (3): 229–33. doi:10.1128/aac.2.3.229. PMC 444296. PMID 4790562.
- ^ Allen LB, Wolf SM, Hintz CJ, Huffman JH, Sidwell RW (March 1977). "Effect of ribavirin on Type 2 Herpesvirus hominis (HVH/2) in vitro and in vivo". Annals of the New York Academy of Sciences. 284 (1): 247–53. Bibcode:1977NYASA.284..247A. doi:10.1111/j.1749-6632.1977.tb21957.x. PMID 212976. S2CID 12159958.
- ^ Allen LB, Cochran KW (November 1972). "Target-Organ Treatment of Neurotropic Virus Disease with Interferon Inducers". Infection and Immunity. 6 (5): 819–23. doi:10.1128/IAI.6.5.819-823.1972. PMC 422616. PMID 4404669.
- ^ Sidwell RW, Huffman JH, Khare GP, Allen LB, Witkowski JT, Robins RK (August 1972). "Broad-spectrum antiviral activity of Virazole: 1-beta-D-ribofuranosyl-1,2,4-triazole-3-carboxamide". Science. 177 (4050): 705–06. Bibcode:1972Sci...177..705S. doi:10.1126/science.177.4050.705. PMID 4340949. S2CID 43106875.
- ^ Babiuk LA, Meldrum B, Gupta VS, Rouse BT (December 1975). "Comparison of the Antiviral Effects of 5-Methoxymethyl-deoxyuridine with 5-Iododeoxyuridine, Cytosine Arabinoside, and Adenine Arabinoside". Antimicrob. Agents Chemother. 8 (6): 643–50. doi:10.1128/aac.8.6.643. PMC 429441. PMID 1239978.
- ^ O'Meara A, Deasy PF, Hillary IB, Bridgen WD (December 1979). "Acyclovir for treatment of mucocutaneous herpes infection in a child with leukaemia". Lancet. 2 (8153): 1196. doi:10.1016/S0140-6736(79)92428-0. PMID 91931. S2CID 40775996.
- ^ Whitley R, Arvin A, Prober C, et al. (February 1991). "A controlled trial comparing vidarabine with acyclovir in neonatal herpes simplex virus infection. Infectious Diseases Collaborative Antiviral Study Group". N. Engl. J. Med. 324 (7): 444–49. doi:10.1056/NEJM199102143240703. PMID 1988829.
- ^ a b c Kimberlin DW, Lin CY, Jacobs RF, et al. (August 2001). "Safety and efficacy of high-dose intravenous acyclovir in the management of neonatal herpes simplex virus infections". Pediatrics. 108 (2): 230–38. doi:10.1542/peds.108.2.230. PMID 11483782.
- ^ Vezina C, Steben M (2001). "Genital Herpes: Psychosexual Impacts and Counselling" (PDF). The Canadian Journal of CME (June): 125–34. Archived from the original (PDF) on 2008-12-16. Retrieved 2008-09-10.
- ^ Green, J; Ferrier, S; Kocsis, A; Shadrick, J; Ukoumunne, OC; Murphy, S; Hetherton, J (February 2003). "Determinants of disclosure of genital herpes to partners". Sexually Transmitted Infections. 79 (1): 42–44. doi:10.1136/sti.79.1.42. PMC 1744583. PMID 12576613.
- ^ Miranda Hitti (24 August 2007). "Stigma Still Strong". WebMD. Archived from the original on 16 November 2013.
- ^ "Herpes groups on Meetup | HerpesDateSites". www.herpesdatesites.com. Archived from the original on 2017-04-06. Retrieved 2017-02-20.
- ^ "Genital Herpes Dating Sites Review | Best Herpes Dating Sites for Genital HSV Singles in 2016". genitalherpesdatingsites.org. Archived from the original on 2017-05-22. Retrieved 2017-02-20.
- ^ "Herpes Support Forum". Archived from the original on 4 May 2016. Retrieved 15 May 2016.
- ^ "Our History". Archived from the original on 21 October 2014. Retrieved 19 October 2014.
ASHA was founded in 1914 in New York City, formed out of early 20th-century social reform movements focused on fighting sexually transmitted infections (known then as venereal disease, or VD) and prostitution.
- ^ "American Social Health Association". Archived from the original on 2008-08-20.
- ^ "The Herpes Resource Center". Archived from the original on 2011-10-03.
- ^ "Helping You With Herpes". Herpes Viruses Association. Archived from the original on 2015-07-26. Retrieved 2017-02-20.
- ^ "mRNA-1608 Herpes Vaccine". www.precisionvaccinations.com. Retrieved 2022-12-11.
- ^ "Can herpes kill you? - Technology Org". www.technology.org. 2022-09-27. Retrieved 2022-12-11.
- ^ Foley, James A. (21 Oct 2013). "Hitchhiking Herpes Virus Aligns with Spread of Human Civilization". NatureWorldNews.com. Archived from the original on 22 October 2013. Retrieved 22 October 2013.
