Homotaurine: Difference between revisions
No edit summary |
|||
| Line 51: | Line 51: | ||
'''Homotaurine''' (also known as '''tramiprosate''' ([[International Nonproprietary Name|INN]]), '''3-amino-1-propanesulfonic acid''', or '''3-APS''') is a natural amino acid found in seaweed.<ref>{{cite journal | pmc = 4172065| year = 2014| last1 = Martorana| first1 = A.| title = Homotaurine Induces Measurable Changes of Short Latency Afferent Inhibition in a Group of Mild Cognitive Impairment Individuals| journal = Frontiers in Aging Neuroscience| volume = 6| pages = 254| last2 = Di Lorenzo| first2 = F.| last3 = Manenti| first3 = G.| last4 = Semprini| first4 = R.| last5 = Koch| first5 = G.| pmid = 25295005| doi = 10.3389/fnagi.2014.00254}}</ref> It is analogous to [[taurine]], but with an extra carbon in its chain. It has [[GABAergic]] activity, apparently by mimicking GABA, which it resembles.<ref name=OrgChem2007/> |
'''Homotaurine''' (also known as '''tramiprosate''' ([[International Nonproprietary Name|INN]]), '''3-amino-1-propanesulfonic acid''', or '''3-APS''') is a natural amino acid found in seaweed.<ref>{{cite journal | pmc = 4172065| year = 2014| last1 = Martorana| first1 = A.| title = Homotaurine Induces Measurable Changes of Short Latency Afferent Inhibition in a Group of Mild Cognitive Impairment Individuals| journal = Frontiers in Aging Neuroscience| volume = 6| pages = 254| last2 = Di Lorenzo| first2 = F.| last3 = Manenti| first3 = G.| last4 = Semprini| first4 = R.| last5 = Koch| first5 = G.| pmid = 25295005| doi = 10.3389/fnagi.2014.00254}}</ref> It is analogous to [[taurine]], but with an extra carbon in its chain. It has [[GABAergic]] activity, apparently by mimicking GABA, which it resembles.<ref name=OrgChem2007/> |
||
Homotaurine was investigated in a [[Phase III clinical trials|Phase III]] clinical trial as a potential treatment for [[Alzheimer's disease]] that did not show efficacy.<ref name=AD2012rev>{{cite journal | doi = 10.3275/8585 | pmid = 22961121 | year = 2012 | last1 = Caltagirone | first1 = C. | title = The potential protective effect of tramiprosate (homotaurine) against Alzheimer's disease: A review | journal = Aging Clinical and Experimental Research | volume = 24 | issue = 6 | pages = 580–7 | last2 = Ferrannini | first2 = L. | last3 = Marchionni | first3 = N. | last4 = Nappi | first4 = G. | last5 = Scapagnini | first5 = G. | last6 = Trabucchi | first6 = M. | s2cid = 10816430 }}</ref> A study in cognitive impairment done in 2018 did show positive benefits.<ref>http://www.jgerontology-geriatrics.com/wp-content/uploads/2018/03/03_Martorana-1.pdf</ref> |
Homotaurine was investigated in a [[Phase III clinical trials|Phase III]] clinical trial as a potential treatment for [[Alzheimer's disease]] that did not show efficacy. However, post-hoc analyses have shown positive and significant effects of homotaurine on secondary endpoints and subgroups of patients, including a reduction in hippocampal volume loss and lower decline in memory function in the overall cohort, as well as a reduction in global cognitive decline in APOE4 allele carriers, suggesting a disease-modifying effects.<ref name=AD2012rev>{{cite journal | doi = 10.3275/8585 | pmid = 22961121 | year = 2012 | last1 = Caltagirone | first1 = C. | title = The potential protective effect of tramiprosate (homotaurine) against Alzheimer's disease: A review | journal = Aging Clinical and Experimental Research | volume = 24 | issue = 6 | pages = 580–7 | last2 = Ferrannini | first2 = L. | last3 = Marchionni | first3 = N. | last4 = Nappi | first4 = G. | last5 = Scapagnini | first5 = G. | last6 = Trabucchi | first6 = M. | s2cid = 10816430 }}</ref> A study in cognitive impairment done in 2018 did show positive benefits.<ref>http://www.jgerontology-geriatrics.com/wp-content/uploads/2018/03/03_Martorana-1.pdf</ref> |
||
Homotaurine is currently in a phase 3 study with expected FDA approval as the first disease modifying drug for AD. <ref>https://pubmed.ncbi.nlm.nih.gov/32787971/</ref> <ref>https://pubmed.ncbi.nlm.nih.gov/29182706/</ref> |
Homotaurine is currently in a phase 3 study with expected FDA approval as the first disease modifying drug for AD. <ref>https://pubmed.ncbi.nlm.nih.gov/32787971/</ref> <ref>https://pubmed.ncbi.nlm.nih.gov/29182706/</ref> |
||
Revision as of 21:55, 13 May 2021
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
3-Aminopropane-1-sulfonic acid | |
| Other names
Tramiprosate; Alzhemed; 3-APS
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.020.889 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C3H9NO3S | |
| Molar mass | 139.17 g·mol−1 |
| Melting point | 293 °C (559 °F; 566 K) (decomposition) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
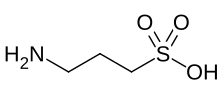
Homotaurine (also known as tramiprosate (INN), 3-amino-1-propanesulfonic acid, or 3-APS) is a natural amino acid found in seaweed.[2] It is analogous to taurine, but with an extra carbon in its chain. It has GABAergic activity, apparently by mimicking GABA, which it resembles.[3]
Homotaurine was investigated in a Phase III clinical trial as a potential treatment for Alzheimer's disease that did not show efficacy. However, post-hoc analyses have shown positive and significant effects of homotaurine on secondary endpoints and subgroups of patients, including a reduction in hippocampal volume loss and lower decline in memory function in the overall cohort, as well as a reduction in global cognitive decline in APOE4 allele carriers, suggesting a disease-modifying effects.[4] A study in cognitive impairment done in 2018 did show positive benefits.[5]
Homotaurine is currently in a phase 3 study with expected FDA approval as the first disease modifying drug for AD. [6] [7]
Biochemical properties
In preclinical studies it had been found to bind to soluble amyloid beta and inhibit the formation of neurotoxic aggregates.[4][8] Homotaurine has also shown anticonvulsant activities, reduction in skeletal muscle tonus, and hypothermic activity.[9]
Homotaurine has been reported as a GABA antagonist,[3] as well as a GABA agonist.[9][10] In vitro studies have found that homotaurine is a GABAA partial agonist[11] as well as a GABAB receptor partial agonist with low efficacy, becoming an antagonist and displacing the full agonists GABA and baclofen at this receptor.[12] In a study in rats, homotaurine reversed the catatonia induced by baclofen (the prototypical GABAB agonist),[13] and was able to produce analgesia via the GABAB receptor, an effect that was abolished when CGP-35348, a GABAB receptor antagonist was applied.[14][15]
In a human study homotaurine selectively and fully inhibits the formation of Aβ42 oligomers at the clinical dose, without evidence of vasogenic edema.[16]
One study in rats showed that homotaurine suppressed ethanol-stimulated dopamine release, as well as ethanol intake and preference in rats in a way similar to the N-acetyl derivative of homotaurine, acamprosate.[17] Acamprosate was approved by the FDA in 2004 to treat alcohol dependence.[3]
References
- ^ "Homotaurine". Sigma-Aldrich.
- ^ Martorana, A.; Di Lorenzo, F.; Manenti, G.; Semprini, R.; Koch, G. (2014). "Homotaurine Induces Measurable Changes of Short Latency Afferent Inhibition in a Group of Mild Cognitive Impairment Individuals". Frontiers in Aging Neuroscience. 6: 254. doi:10.3389/fnagi.2014.00254. PMC 4172065. PMID 25295005.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ a b c Lednicer D (2008). The Organic Chemistry of Drug Synthesis (7th ed.). Hoboken: John Wiley & Sons. p. 15. ISBN 978-0-470-18066-2.
- ^ a b Caltagirone, C.; Ferrannini, L.; Marchionni, N.; Nappi, G.; Scapagnini, G.; Trabucchi, M. (2012). "The potential protective effect of tramiprosate (homotaurine) against Alzheimer's disease: A review". Aging Clinical and Experimental Research. 24 (6): 580–7. doi:10.3275/8585. PMID 22961121. S2CID 10816430.
- ^ http://www.jgerontology-geriatrics.com/wp-content/uploads/2018/03/03_Martorana-1.pdf
- ^ https://pubmed.ncbi.nlm.nih.gov/32787971/
- ^ https://pubmed.ncbi.nlm.nih.gov/29182706/
- ^ Aisen, Paul; Gauthier, Serge; Vellas, Bruno; Briand, Richard; Saumier, Daniel; Laurin, Julie; Garceau, Denis (2007). "Alzhemed: A Potential Treatment for Alzheimers Disease". Current Alzheimer Research. 4 (4): 473–478. doi:10.2174/156720507781788882. PMID 17908052.
- ^ a b Oja SS and Kontro P. (2013). Lajtha ANS (ed.). Chapter 18: Taurine. Springer Science & Business Media. p. 520. ISBN 9781468443677.
{{cite book}}:|work=ignored (help) - ^ Armen H. Tashjian and Ehrin J. Armstrong. Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy. Edited by David E. Golan. Lippincott Williams & Wilkins, 2011 ISBN 9781451118056. Page 308
- ^ Reyes-Haro, Daniel; Cabrera-Ruíz, Elizabeth; Estrada-Mondragón, Argel; Miledi, Ricardo; Martínez-Torres, Ataúlfo (2014). "Modulation of GABA-A receptors of astrocytes and STC-1 cells by taurine structural analogs". Amino Acids. 46 (11): 2587–2593. doi:10.1007/s00726-014-1813-0. PMID 25119985. S2CID 10319072.
- ^ Giotti, A.; Luzzi, S.; Spagnesi, S.; Zilletti, Lucilla (1983). "Homotaurine: A GABAB antagonist in guinea-pig ileum". British Journal of Pharmacology. 79 (4): 855–862. doi:10.1111/j.1476-5381.1983.tb10529.x. PMC 2044932. PMID 6652358.
- ^ Mehta, A.; Ticku, M. (1987). "Baclofen induces catatonia in rats". Neuropharmacology. 26 (9): 1419–1423. doi:10.1016/0028-3908(87)90108-0. PMID 2823166. S2CID 24010833.
- ^ Serrano, M.Isabel; Serrano, Jose S.; Fernández, Ana; Asadi, Ihklas; Serrano-Martino, M.Carmen (1998). "GABAB Receptors and Opioid Mechanisms Involved in Homotaurine-Induced Analgesia". General Pharmacology: The Vascular System. 30 (3): 411–415. doi:10.1016/s0306-3623(97)00279-6. PMID 9510095.
- ^ Serrano, Maria Isabel; Serrano, Jose S.; Asadi, Ikhlas; Fernandez, Ana; Serrano-Martino, Maria Carmen (2001). "Role of K+-channels in homotaurine-induced analgesia". Fundamental and Clinical Pharmacology. 15 (3): 167–173. doi:10.1046/j.1472-8206.2001.00026.x. PMID 11468027. S2CID 19694376.
- ^ https://pubmed.ncbi.nlm.nih.gov/32787971/
- ^ Olive, M.Foster; Nannini, Michelle A.; Ou, Christine J.; Koenig, Heather N.; Hodge, Clyde W. (2002). "Effects of acute acamprosate and homotaurine on ethanol intake and ethanol-stimulated mesolimbic dopamine release". European Journal of Pharmacology. 437 (1–2): 55–61. doi:10.1016/s0014-2999(02)01272-4. PMID 11864639.
