Free radical damage to DNA: Difference between revisions
Citation bot (talk | contribs) Alter: template type. Add: pmid, doi-access. | Use this bot. Report bugs. | Suggested by Headbomb | Linked from Wikipedia:WikiProject_Academic_Journals/Journals_cited_by_Wikipedia/Sandbox | #UCB_webform_linked 811/910 |
m link ionizing radiation using Find link |
||
| Line 1: | Line 1: | ||
{{see also|DNA damage (naturally occurring)}} |
{{see also|DNA damage (naturally occurring)}} |
||
'''Free radical damage to DNA''' can occur as a result of exposure to |
'''Free radical damage to DNA''' can occur as a result of exposure to [[ionizing radiation]] or to radiomimetic<ref name="controversy" /> compounds. Damage to [[DNA]] as a result of [[free radical]] attack is called [[indirect DNA damage]] because the radicals formed can diffuse throughout the body and affect other organs. [[Malignant melanoma]] can be caused by indirect DNA damage because it is found in parts of the body not exposed to sunlight. DNA is vulnerable to radical attack because of the very [[Hydrogen atom abstraction|labile hydrogens]] that can be abstracted and the prevalence of double bonds in the [[DNA bases]] that [[Radical addition|radicals can easily add]] to.<ref>{{cite journal | vauthors = Greenberg MM | title = Reactivity of Nucleic Acid Radicals | journal = Advances in Physical Organic Chemistry | volume = 50 | pages = 119–202 | date = 2016 | pmid = 28529390 | pmc = 5435387 | doi = 10.1016/bs.apoc.2016.02.001 | publisher = Elsevier | isbn = 978-0-12-804716-3 }}</ref> |
||
==Damage via radiation exposure== |
==Damage via radiation exposure== |
||
Revision as of 11:24, 2 November 2021
Free radical damage to DNA can occur as a result of exposure to ionizing radiation or to radiomimetic[1] compounds. Damage to DNA as a result of free radical attack is called indirect DNA damage because the radicals formed can diffuse throughout the body and affect other organs. Malignant melanoma can be caused by indirect DNA damage because it is found in parts of the body not exposed to sunlight. DNA is vulnerable to radical attack because of the very labile hydrogens that can be abstracted and the prevalence of double bonds in the DNA bases that radicals can easily add to.[2]
Damage via radiation exposure
Radiolysis of intracellular water by ionizing radiation creates peroxides, which are relatively stable precursors to hydroxyl radicals. 60%- 70% of cellular DNA damage is caused by hydroxyl radicals,[3] yet hydroxyl radicals are so reactive that they can only diffuse one or two molecular diameters before reacting with cellular components. Thus, hydroxyl radicals must be formed immediately adjacent to nucleic acids in order to react. Radiolysis of water creates peroxides that can act as diffusable, latent forms of hydroxyl radicals. Some metal ions in the vicinity of DNA generate the hydroxyl radicals from peroxide.[4]
- H2O + hν → H2O+ + e−
- H2O + e− → H2O−
- H2O+ → H+ + OH·
- H2O− → OH− + H·
- 2 OH· →H2O2
Free radical damage to DNA is thought to result in mutations that may lead to some cancers.
The Fenton Reaction
The Fenton reaction results in the creation of hydroxyl radicals from hydrogen peroxide and an Iron (II) catalyst. Iron(III) is regenerated via the Haber-Weiss reaction. Transition metals with a free coordination site are capable of reducing peroxides to hydroxyl radicals.[1] Iron is believed to be the metal responsible for the creation of hydroxyl radicals because it exists at the highest concentration of any transition metal in most living organisms.[5] The Fenton reaction is possible because transition metals can exist in more than one oxidation state and their valence electrons may be unpaired, allowing them to participate in one-electron redox reactions.
- Fe2+ + H2O2 → Fe3+ + OH· + OH−
The creation of hydroxyl radicals by iron(II) catalysis is important because iron(II) can be found coordinated with, and therefore in close proximity to, DNA. This reaction allows for hydrogen peroxide created by radiolysis of water to diffuse to the nucleus and react with Iron (II) to produce hydroxyl radicals, which in turn react with DNA. The location and binding of Iron (II) to DNA may play an important role in determining the substrate and nature of the radical attack on the DNA. The Fenton reaction generates two types of oxidants, Type I and Type II. Type I oxidants are moderately sensitive to peroxides and ethanol.[5] Type I and Type II oxidants preferentially cleave at the specific sequences.[5]
Radical hydroxyl attack

Hydroxyl radicals can attack the deoxyribose DNA backbone and bases, potentially causing a plethora of lesions that can be cytotoxic or mutagenic. Cells have developed complex and efficient repair mechanisms to fix the lesions. In the case of free radical attack on DNA, base-excision repair is the repair mechanism used. Hydroxyl radical reactions with the deoxyribose sugar backbone are initiated by hydrogen abstraction from a deoxyribose carbon, and the predominant consequence is eventual strand breakage and base release. The hydroxyl radical reacts with the various hydrogen atoms of the deoxyribose in the order 5′ H > 4′ H > 3′ H ≈ 2′ H ≈ 1′ H. This order of reactivity parallels the exposure to solvent of the deoxyribose hydrogens.[6]
Hydroxyl radicals react with DNA bases via addition to the electron-rich, pi bonds. These pi bonds in the bases are located between C5-C6 of pyrimidines and N7-C8 in purines.[7] Upon addition of the hydroxyl radical, many stable products can be formed. In general, radical hydroxyl attacks on base moieties do not cause altered sugars or strand breaks except when the modifications labilize the N-glycosyl bond, allowing the formation of baseless sites that are subject to beta-elimination.
Abasic sites
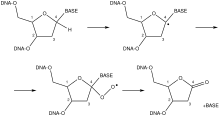
Hydrogen abstraction from the 1’-deoxyribose carbon by the hydroxyl radical creates a 1 ‘-deoxyribosyl radical. The radical can then react with molecular oxygen, creating a peroxyl radical which can be reduced and dehydrated to yield a 2’-deoxyribonolactone and free base. A deoxyribonolactone is mutagenic and resistant to repair enzymes. Thus, an abasic site is created.[8]
Radical damage through radiomimetic compounds
Radical damage to DNA can also occur through the interaction of DNA with certain natural products known as radiomimetic compounds, molecular compounds which affect DNA in similar ways to radiation exposure. Radiomimetic compounds induce double-strand breaks in DNA via highly specific, concerted free-radical attacks on the deoxyribose moieties in both strands of DNA.
General mechanism
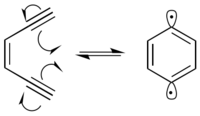
Many radiomimetic compounds are enediynes, which undergo the Bergman cyclization reaction to produce a 1,4-didehydrobenzene diradical. The 1,4-didehydrobenzene diradical is highly reactive, and will abstract hydrogens from any possible hydrogen-donor.
In the presence of DNA, the 1,4-didehydrobenzene diradical abstracts hydrogens from the deoxyribose sugar backbone, predominantly at the C-1’, C-4’ and C-5’ positions. Hydrogen abstraction causes radical formation at the reacted carbon. The carbon radical reacts with molecular oxygen, which leads to a strand break in the DNA through a variety of mechanisms.[9] 1,4-Didehydrobenzene is able to position itself in such a way that it can abstract proximal hydrogens from both strands of DNA.[10] This produces a double-strand break in the DNA, which can lead to cellular apoptosis if not repaired.
Enediynes generally undergo the Bergman cyclization at temperatures exceeding 200 °C. However, incorporating the enediyne into a 10-membered cyclic hydrocarbon makes the reaction more thermodynamically favorable by releasing the ring strain of the reactants. This allows for the Bergman cyclization to occur at 37 °C, the biological temperature of humans. Molecules which incorporate enediynes into these larger ring structures have been found to be extremely cytotoxic.[11]
Natural products
Enediynes are present in many complicated natural products. They were originally discovered in the early 1980s during a search for new anticancer products produced by microorganisms.[10] Calicheamicin was one of the first such products identified and was originally found in a soil sample taken from Kerrville, Texas. These compounds are synthesized by bacteria as defense mechanisms due to their ability to cleave DNA through the formation of 1,4-didehydrobenzene from the enediyne component of the molecule.
Calicheamicin and other related compounds share several common characteristics. The extended structures attached to the enediyne allow the compound to specifically bind DNA,[12] in most cases to the minor groove of the double helix. Additionally, part of the molecule is known as the “trigger” which, under specific physiological conditions, activates the enediyne, known as the “warhead” and 1,4-didehydrobenzene is generated.
Three classes of enediynes have since been identified: calicheamicin, dynemicin, and chromoprotein based products.
The calicheamicin types are defined by a methyl trisulfide group that is involved in triggering the molecule by the following mechanism.[10]
Calicheamicin and the closely related esperamicin have been used as anticancer drugs due to their high toxicity and specificity.[10]
Dynemicin and its relatives are characterized by the presence of an anthraquinone and enediyne core. The anthraquinone component allows for specific binding of DNA at the 3’ side of purine bases through intercalation, a site that is different from calicheamicin. Its ability to cleave DNA is greatly increased in the presence of NADPH and thiol compounds.[13] This compound has also found prominence as an antitumor agent.[13]
Chromoprotein enediynes are characterized by an unstable chromophore enediyne bound to an apoprotein.
The chromophore is unreactive when bound to the apoprotein. Upon its release, it reacts to form 1,4-didehydrobenzene and subsequently cleaves DNA.
Antitumor ability
Most enediynes, including the ones listed above, have been used as potent antitumor antibiotics due to their ability to efficiently cleave DNA. Calicheamicin and esperamicin are the two most commonly used types due to their high specificity when binding to DNA, which minimizes unfavorable side reactions.[12] They have been shown to be especially useful for treating acute myeloid leukemia.[14]
Additionally, calicheamicin is able to cleave DNA at low concentrations, proving to be up to 1000 times more effective than adriamycin at combating certain types of tumors.[15] In all cases, cells lack the ability to repair double-stranded DNA breaks, making these compounds especially effective for treating tumor cells.
The free radical mechanism to treat certain types of cancers extends beyond enediynes. Tirapazamine generates a free radical under anoxic conditions instead of the trigger mechanism of an enediyne. The free radical then continues on to cleave DNA in a similar manner to 1,4-didehydrobenzene in order to treat cancerous cells. It is currently in Phase III trials.
References
- ^ a b Barbusinski K (2009). "Fenton Reaction- Controversy Concerning the Chemistry". Ecological Chemistry and Engineering. 16 (3).
- ^ Greenberg MM (2016). "Reactivity of Nucleic Acid Radicals". Advances in Physical Organic Chemistry. 50. Elsevier: 119–202. doi:10.1016/bs.apoc.2016.02.001. ISBN 978-0-12-804716-3. PMC 5435387. PMID 28529390.
- ^ Ward JF (1988). DNA damage produced by ionizing radiation in mammalian cells: identities, mechanisms of formation, and reparability. Progress in Nucleic Acid Research and Molecular Biology. Vol. 35. pp. 95–125. doi:10.1016/s0079-6603(08)60611-x. ISBN 9780125400350. PMID 3065826.
- ^ Henle ES, Linn S (August 1997). "Formation, prevention, and repair of DNA damage by iron/hydrogen peroxide". The Journal of Biological Chemistry. 272 (31): 19095–8. doi:10.1074/jbc.272.31.19095. PMID 9235895. S2CID 11016259.
- ^ a b c Pogozelski WK, Tullius TD (May 1998). "Oxidative Strand Scission of Nucleic Acids: Routes Initiated by Hydrogen Abstraction from the Sugar Moiety". Chemical Reviews. 98 (3): 1089–1108. doi:10.1021/cr960437i. PMID 11848926.
- ^ Balasubramanian B, Pogozelski WK, Tullius TD (August 1998). "DNA strand breaking by the hydroxyl radical is governed by the accessible surface areas of the hydrogen atoms of the DNA backbone". Proceedings of the National Academy of Sciences of the United States of America. 95 (17): 9738–43. Bibcode:1998PNAS...95.9738B. doi:10.1073/pnas.95.17.9738. PMC 21406. PMID 9707545.
- ^ Steenken S (1989). "Purine bases, nuclesides and nucleotides: aqueous solution redox chemistry and transformation reactions of their radical cations and e- and OH adducts". Chem. Rev. 89 (3): 503–529. doi:10.1021/cr00093a003.
- ^ Lhomme J, Constant JF, Demeunynck M (1999). "Abasic DNA structure, reactivity, and recognition". Biopolymers. 52 (2): 65–83. doi:10.1002/1097-0282(1999)52:2<65::aid-bip1>3.3.co;2-l. PMID 10898853.
- ^ Povirk LF (1996). "DNA damage and mutagenesis by radiomimetic DNA-cleaving agents: Bleomycin, neocarzinostatin and other enediynes". Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis. 355 (1–2): 71–89. doi:10.1016/0027-5107(96)00023-1. PMID 8781578.
- ^ a b c d Kraka E, Cremer D (2000). "Computer design of anticancer drugs. A new enediyne warhead". J. Am. Chem. Soc. 122 (34): 8245–8264. doi:10.1021/ja001017k.
- ^ Zhen YS, Ming XY, Yu B, Otani T, Saito H, Yamada Y (August 1989). "A new macromolecular antitumor antibiotic, C-1027. III. Antitumor activity". The Journal of Antibiotics. 42 (8): 1294–8. doi:10.7164/antibiotics.42.1294. PMID 2759910.
- ^ a b Ellestad GA (September 2011). "Structural and conformational features relevant to the anti-tumor activity of calicheamicin γ 1I". Chirality. 23 (8): 660–71. doi:10.1002/chir.20990. PMID 21800378.
- ^ a b Sugiura Y, Shiraki T, Konishi M, Oki T (May 1990). "DNA intercalation and cleavage of an antitumor antibiotic dynemicin that contains anthracycline and enediyne cores". Proceedings of the National Academy of Sciences of the United States of America. 87 (10): 3831–5. Bibcode:1990PNAS...87.3831S. doi:10.1073/pnas.87.10.3831. PMC 53997. PMID 2339123.
- ^ Sievers EL, Appelbaum FR, Spielberger RT, Forman SJ, Flowers D, Smith FO, Shannon-Dorcy K, Berger MS, Bernstein ID (June 1999). "Selective ablation of acute myeloid leukemia using antibody-targeted chemotherapy: a phase I study of an anti-CD33 calicheamicin immunoconjugate". Blood. 93 (11): 3678–84. doi:10.1182/blood.V93.11.3678. PMID 10339474.
- ^ Zein N, Sinha AM, McGahren WJ, Ellestad GA (May 1988). "Calicheamicin gamma 1I: an antitumor antibiotic that cleaves double-stranded DNA site specifically". Science. 240 (4856): 1198–201. Bibcode:1988Sci...240.1198Z. doi:10.1126/science.3240341. PMID 3240341.