Calcium hypochlorite: Difference between revisions
Gopal Tamili (talk | contribs) () removed Tags: Mobile edit Mobile web edit |
No edit summary |
||
| Line 67: | Line 67: | ||
==Uses== |
==Uses== |
||
===History=== |
|||
[[Charles Tennant]] and [[Charles Macintosh]] developed an industrial process for the manufactured of Chloride of Lime in the late 18th Century.<ref>{{cite web|url=https://www.chemistryworld.com/podcasts/calcium-hypochlorite/3008985.article |title=Calcium hypochlorite |publisher=Chemistry World}}</ref> It was patented in 1799. |
|||
===Sanitation=== |
===Sanitation=== |
||
Calcium hypochlorite is commonly used to [[swimming pool sanitation|sanitize public swimming pools]] and [[water treatment|disinfect drinking water]]. Generally the commercial substances are sold with a purity of 65% to 73% with other chemicals present, such as calcium chloride and calcium carbonate, resulting from the manufacturing process. As a swimming pool chemical, it is blended with other chemicals less often than other forms of chlorine, due to dangerous reactions with some common pool chemicals. In solution, calcium hypochlorite could be used as a general purpose sanitizer,<ref>{{cite techreport|institution=Mannsvile Chemical Products |date=1987 |title=Chemical Products Synopsis: Calcium Hypochlorite |location=Asbuiy Park, NJ}}</ref> but due to calcium residue, sodium hypochlorite (bleach) is usually preferred. |
Calcium hypochlorite is commonly used to [[swimming pool sanitation|sanitize public swimming pools]] and [[water treatment|disinfect drinking water]]. Generally the commercial substances are sold with a purity of 65% to 73% with other chemicals present, such as calcium chloride and calcium carbonate, resulting from the manufacturing process. As a swimming pool chemical, it is blended with other chemicals less often than other forms of chlorine, due to dangerous reactions with some common pool chemicals. In solution, calcium hypochlorite could be used as a general purpose sanitizer,<ref>{{cite techreport|institution=Mannsvile Chemical Products |date=1987 |title=Chemical Products Synopsis: Calcium Hypochlorite |location=Asbuiy Park, NJ}}</ref> but due to calcium residue, sodium hypochlorite (bleach) is usually preferred. |
||
Revision as of 20:05, 15 October 2020
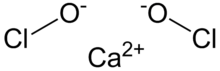
| |
| Names | |
|---|---|
| Other names
Hypochlorous acid calcium salt, bleaching powder, calcium oxychloride, chloride of lime
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.029.007 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1748 2208 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| CaO(Cl)2 | |
| Molar mass | 142.98 g/mol |
| Appearance | white/gray powder |
| Density | 2.35 g/cm3 (20 °C) |
| Melting point | 100 °C (212 °F; 373 K) |
| Boiling point | 175 °C (347 °F; 448 K) decomposes |
| 21 g/100 mL at 25 °C | |
| Solubility | reacts in alcohol |
| Hazards | |
| GHS labelling: | |
   
| |
| Danger | |
| H272, H302, H314, H400 | |
| P210, P220, P221, P260, P264, P270, P273, P280, P301+P312, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P330, P363, P370+P378, P391, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
850 mg/kg (oral, rat) |
| Safety data sheet (SDS) | ICSC 0638 |
| Related compounds | |
Other anions
|
Calcium chloride |
Other cations
|
Sodium hypochlorite |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Calcium hypochlorite is an inorganic compound with formula CaO(Cl)2. It is the main active ingredient of commercial products called bleaching powder, chlorine powder, or chlorinated lime, used for water treatment and as a bleaching agent.[1] This compound is relatively stable and has greater available chlorine than sodium hypochlorite (liquid bleach).[2] It is a white solid, although commercial samples appear yellow. It strongly smells of chlorine, owing to its slow decomposition in moist air. It is not highly soluble in hard water, and is more preferably used in soft to medium-hard water. It has two forms: dry (anhydrous); and hydrated (hydrous).
Uses
History
Charles Tennant and Charles Macintosh developed an industrial process for the manufactured of Chloride of Lime in the late 18th Century.[3] It was patented in 1799.
Sanitation
Calcium hypochlorite is commonly used to sanitize public swimming pools and disinfect drinking water. Generally the commercial substances are sold with a purity of 65% to 73% with other chemicals present, such as calcium chloride and calcium carbonate, resulting from the manufacturing process. As a swimming pool chemical, it is blended with other chemicals less often than other forms of chlorine, due to dangerous reactions with some common pool chemicals. In solution, calcium hypochlorite could be used as a general purpose sanitizer,[4] but due to calcium residue, sodium hypochlorite (bleach) is usually preferred.
Organic chemistry
Calcium hypochlorite is a general oxidizing agent and therefore finds some use in organic chemistry.[5] For instance the compound is used to cleave glycols, α-hydroxy carboxylic acids and keto acids to yield fragmented aldehydes or carboxylic acids.[6] Calcium hypochlorite can also be used in the haloform reaction to manufacture chloroform.[7] Calcium hypochlorite can be used to oxidize thiol and sulfide byproducts in organic synthesis and thereby reduce their odour and make them safe to dispose of.[8]
Production
Calcium hypochlorite is produced industrially by treating lime (Ca(OH)2) with chlorine gas. The reaction can be conducted in stages to give various compositions, each with different concentration of calcium hypochlorite, together with unconverted lime and calcium chloride. The full conversion is shown[1]
- 2 Cl
2 + 2 Ca(OH)
2 → Ca(ClO)
2 + CaCl
2 + 2 H
2O
Bleaching powder is made with slightly moist slaked lime. It is not a simple mixture of calcium hypochlorite, calcium chloride, and calcium hydroxide. Instead, it is a mixture consisting principally of calcium hypochlorite Ca(ClO)2, dibasic calcium hypochlorite, Ca3(ClO)2(OH)4 (also written as Ca(ClO)2 · 2 Ca(OH)2), and dibasic calcium chloride, Ca3Cl2(OH)4 (calcium hydroxychloride also written as CaCl2 · 2 Ca(OH)2).[9]
Calcium oxychlorides
A confusion sometimes reigns between calcium oxychlorides and calcium hypochlorite. Indeed, the name calcium oxychloride (or calcium hydroxychloride) does not immediately refer to calcium hypochlorite, but is only applicable to the mixed calcium basic chloride compounds remaining unreacted in the bleaching powder, such as, e.g. CaCl2 · 2 Ca(OH)2.
Calcium oxychloride may also be formed in concrete in roads and bridges when calcium chloride is used as deicing agent during winter. Calcium chloride then reacts with calcium hydroxide (portlandite) present in cement hydration products and forms a deleterious expanding phase also named CAOXY (abbreviation for calcium oxychloride) by concrete technologists. The stress induced into concrete by crystallisation pressure and CAOXY salt expansion can considerably reduce the strength of concrete.[10][11]
Safety
Calcium hypochlorite is strongly basic. It is also a strong oxidizing agent, as it contains a chlorine atom at the valence I (redox state: Cl+1).
Calcium hypochlorite is stored dry and cold, away from any acid, organic materials, and metals. The hydrated form is safer to handle.
References
- ^ a b Vogt, H.; Balej, J; Bennett, J. E.; Wintzer, P.; Sheikh, S. A.; Gallone, P.; Vasudevan, S.; Pelin, K. (2010). "Chlorine Oxides and Chlorine Oxygen Acids". Ullmann's Encyclopedia of Industrial Chemistry. Wiley-VCH. doi:10.1002/14356007.a06_483.pub2. ISBN 978-3527306732.
- ^ Gerald F. Connell. "Key operating strategies for chlorine disinfection operating systems" (PDF). Retrieved 19 October 2014.
- ^ "Calcium hypochlorite". Chemistry World.
- ^ Chemical Products Synopsis: Calcium Hypochlorite (Technical report). Asbuiy Park, NJ: Mannsvile Chemical Products. 1987.
- ^ Nwaukwa, Stephen; Keehn, Philip (1982). "The oxidation of aldehydes to acids with calcium hypochlorite [Ca(OCl)2]". Tetrahedron Letters. 23 (31): 3131–3134. doi:10.1016/S0040-4039(00)88577-9.
- ^ Nwaukwa, Stephen; Keehn, Philip (1982). "Oxidative cleavage of α-diols, α-diones, α-hydroxy-ketones and α-hydroxy- and α-keto acids with calcium hypochlorite [Ca(OCl)2]". Tetrahedron Letters. 23 (31): 3135–3138. doi:10.1016/S0040-4039(00)88578-0.
- ^ Cohen, Julius (1900). Practical Organic Chemistry for Advanced Students. New York: Macmillan & Co. p. 63.
- ^ National Research Council (1995). Prudent Practices in the Laboratory: Handling and Disposal of Chemicals. Washington, DC: The National Academies Press. p. 161. doi:10.17226/4911. ISBN 978-0-309-05229-0.
{{cite book}}: External link in|ref= - ^ W.L Smith, Inorganic bleaches, Production of Hypochlorite in Handbook of Detergents,Part F, (2009) Ed. U Zoller and Paul Sosis, CRC Press, ISBN 978-0-8247-0349-3
- ^ "Calcium-munching bacteria could be a secret weapon against road salt eating away at concrete roads and bridges". The Conversation. Retrieved 7 April 2019.
- ^ Suraneni, Prannoy; Monical, Jonathan; Unal, Erol; Farnam, Yaghoob; Weiss, Jason (2017). "Calcium Oxychloride Formation Potential in Cementitious Pastes Exposed to Blends of Deicing Salt". ACI Materials Journal. 114 (4). doi:10.14359/51689607.

