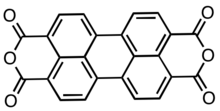
Perylenetetracarboxylic dianhydride
| |

| |
| Names | |
|---|---|
| Other names
Perylene-3,4,9,10-tetracarboxylic dianhydride, Pigment Red 224
| |
| Identifiers | |
| ECHA InfoCard | 100.004.461 |
CompTox Dashboard (EPA)
|
|
| Properties | |
| C24H8O6 | |
| Molar mass | 392.32 |
| Density | 1.7 g/cm3 |
| Melting point | ~350 °C[1] |
| Structure | |
| Monoclinic, P21/c | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Perylenetetracarboxylic dianhydride (PTCDA) is an organic dye molecule and an organic semiconductor. It is used as a precursor to a class of molecules known as Rylene dyes, which are useful as pigments, dyes and organic semiconductors.
Structure
PTCDA consists of a perylene core to which two anhydride groups have been attached, one at either side. It occurs in two crystalline forms, α and β[2]. Both have the P21/c monoclinic symmetry and a density of ca. 1.7 g/cm3, which is relatively high for organic compounds. Their lattice parameters are:
| Form | a | b | c | γ |
|---|---|---|---|---|
| α | 0.374 nm | 1.196 nm | 1.734 nm | 98.8° |
| β | 0.378 nm | 1.930 nm | 1.077 nm | 83.6° |
Uses


As a chemical precursor
The main industrial use of PTCDA is as a precursor to Rylene dyes.[5][6]
As an organic semiconductor
PTCDA's relatively simple chemical structure makes it attractive as a model compound for the study of organic semiconductors. It has among others been used to create single-crystal OFETs[7] and organic solar cells[8]. It is also often used in the study of the interaction between organic semiconductors and the surfaces of metals[9], insulators[4] and inorganic semiconductors[3]. The application of PTCDA as an organic semiconductor is limited by it's low solubility in common solvents[10], which makes it unattractive for most industrial applications.
References
- ^ PTCDA.
- ^ Möbus, M. and Karl, N. (1992). "Structure of perylene-tetracarboxylic-dianhydride thin films on alkali halide crystal substrates". Journal of Crystal Growth. 116 (3–4): 495–504. doi:10.1016/0022-0248(92)90658-6.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Iwata, Kota; Yamazaki, Shiro; Mutombo, Pingo; Hapala, Prokop; Ondráček, Martin; Jelínek, Pavel; Sugimoto, Yoshiaki (2015). "Chemical structure imaging of a single molecule by atomic force microscopy at room temperature". Nature Communications. 6: 7766. doi:10.1038/ncomms8766. PMC 4518281. PMID 26178193.
- ^ a b Cochrane, K. A.; Schiffrin, A.; Roussy, T. S.; Capsoni, M.; Burke, S. A. (2015). "Pronounced polarization-induced energy level shifts at boundaries of organic semiconductor nanostructures". Nature Communications. 6: 8312. doi:10.1038/ncomms9312. PMC 4600718. PMID 26440933.
- ^ Hunger, K. and Herbst, W. (2012) "Pigments, Organic" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim. doi:10.1002/14356007.a20_371
- ^ Greene, M. (2009) "Perylene Pigments", pp. 261–274 in High Performance Pigments, Wiley-VCH, Weinheim.doi:10.1002/9783527626915.ch16
- ^ Nguyen, Linh-Nam; Kumar Pradhan, Sunil; Yen, Chia-Nan; Lin, Ming-Chou; Chen, Chien-Han; Wu, Cen-Shawn; Chang-Liao, Kuei-Shu; Lin, Minn-Tsong; Chen, Chii-Dong (2013). "High performance phototransistors based on single crystalline perylene-tetracarboxylic-dianhydride nanoparticle". Applied Physics Letters. 103 (18): 183301. doi:10.1063/1.4827975. ISSN 0003-6951.
- ^ Kumar, Lokendra; Chaudhary, Dhirendra K. (2014). "Studies on Photovoltaic Properties of ZnPc/PTCDA Based Bilayer Organic Solar Cells". Advanced Science Letters. 20 (7): 1515–1518. doi:10.1166/asl.2014.5728. ISSN 1936-6612.
- ^ Tautz, F.S. (2007). "Structure and bonding of large aromatic molecules on noble metal surfaces: The example of PTCDA". Progress in Surface Science. 82 (9–12): 479–520. doi:10.1016/j.progsurf.2007.09.001. ISSN 0079-6816.
- ^ Russell, James C.; Blunt, Matthew O.; Goretzki, Gudrun; Phillips, Anna G.; Champness, Neil R.; Beton, Peter H. (2010). "Solubilized Derivatives of Perylenetetracarboxylic Dianhydride (PTCDA) Adsorbed on Highly Oriented Pyrolytic Graphite". Langmuir. 26 (6): 3972–3974. doi:10.1021/la903335v. ISSN 0743-7463.

