2-Ethylanthraquinone
| |
| |
| Names | |
|---|---|
| Other names
2-Ethyl-9,10-anthracenedione
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.396 |
| EC Number |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C16H12O2 | |
| Molar mass | 236.27 g/mol |
| Appearance | white to yellowish crystals or powder |
| Density | 1.231g/cm3 |
| Melting point | 105 °C (221 °F; 378 K) |
| Boiling point | 415.4 @ 760mmHg |
| Hazards | |
| Flash point | 155.4 °C, |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
2-Ethylanthraquinone also called2-Ethyl-9,10-anthraquinone is an aromatic organic compound with the formula C16H12O2. The slightly yellow powder is used in the production of hydrogen peroxide (H2O2).
Production
2-Ethylanthraquinone is prepared from the reaction of phthalic anhydride and ethylbenzene: C6H4(CO)2O + C6H5CH2CH3 → C6H4C2O2C6H3C2H5 + H2O. Both phthalic anhydride and ethylbenzene are readily available but are mainly used in the large-scale production of plastics.
Uses
Hydrogen peroxide is produced industrially by the anthraquinone process which involves using 2-alkyl-9,10-anthraquinones for hydrogenation. Many derivatives of anthraquinone are used but 2-Ethylanthraquinone is common because of its high selectivity. The hydrogenation of the unsubsituted ring can reach 90% selectivity by using 2-ethylanthraquinone. Hydrogenation follows the Riedl-Pfleiderer, or autoxidation, process:
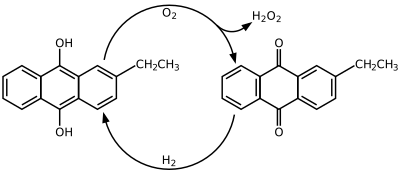
The hydrogenation of 2-ethylanthrahydroquinone is catalyzed by palladium metal. During hydrogenation both 2-ethylanthrahydroquinone and tetrahydroanthraquinone are produced. The tetrahydro derivative of 2-alkylanthraquinone is easily hyrdrogenated but is more difficult to oxidize. This has led to a split in production methods, with current research focusing on the effectiveness of the tetrahydro system. The formation of the tetrahyrdo derivative can be suppressed through use of special catalysts, solvents, and conditions. A working solution must be able to keep the 2-ethylanthraquinone dissolved in the hyrdrogenation, oxidation, and extraction steps. Since hydroquinones dissolve better in polar solvents, and quinones dissolve better in aromatic nonpolar solvents, a solvent mixture must be used. Some suggested mixtures are polyalkylated benzenes and alkyl phosphates or tetraalkyl ureas, trimethylbenzenes and alkylcyclohexanol esters, and methylnaphthalene and nonyl alcohols.
