2-Ethoxyethanol
Appearance
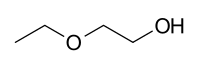
| |
| Names | |
|---|---|
| IUPAC name
2-ethoxyethanol
| |
| Other names
Cellosolve
ethylene glycol ethyl ether oxitol Ethyl Cellosolve | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.003.459 |
| KEGG | |
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H10O2 | |
| Molar mass | 90.122 g·mol−1 |
| Appearance | clear liquid |
| Density | 0.930 g/cm3, liquid |
| Melting point | −70 °C (−94 °F; 203 K) |
| Boiling point | 135 °C (275 °F; 408 K) |
| miscible | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | 44 °C |
| Related compounds | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
2-Ethoxyethanol, also known by the trademark Cellosolve or ethyl cellosolve, is a solvent used widely in commercial and industrial applications. It is a clear, colorless, nearly odorless liquid that is miscible with water, ethanol, diethyl ether, acetone, and ethyl acetate. [1]
2-Ethoxyethanol can be manufactured by the reaction of ethylene oxide with ethanol.
As with other glycol ethers, 2-ethoxyethanol has the useful property of being able to dissolve chemically diverse compounds. It will dissolve oils, resins, grease, waxes, nitrocellulose, and lacquers. This is an ideal property as a multi-purpose cleaner, and, therefore, 2-ethoxyethanol is used in products, such as varnish removers and degreasing solutions.
References
- ^ National Research Council (U.S.). Subcommittee on Spacecraft Maximum Allowable Concentrations (1996). Spacecraft maximum allowable concentrations for selected airborne contaminants. National Academies Press. p. 189. ISBN 978-0-309-05478-2. Retrieved 19 February 2012.

