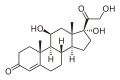
Loteprednol
 | |
| Clinical data | |
|---|---|
| Trade names | Lotemax |
| Other names | HGP 1 |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Routes of administration | Eye drops |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | None |
| Protein binding | 95% |
| Metabolism | Ester hydrolysis |
| Metabolites | Δ1-cortienic acid and its etabonate |
| Onset of action | ≤2 hrs (allergic conjunctivitis) |
| Elimination half-life | 2.8 hrs |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.167.120 |
| Chemical and physical data | |
| Formula | C24H31ClO7 |
| Molar mass | 466.951 g/mol g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 220.5 to 223.5 °C (428.9 to 434.3 °F) |
| Solubility in water | 0.0005 mg/mL (20 °C) |
| |
| |
| | |
Loteprednol (as the ester loteprednol etabonate) is a corticosteroid used to treat inflammations of the eye. It is marketed by Bausch and Lomb as Lotemax.[1]
Medical uses
Applications for this drug include the reduction of inflammation after eye surgery,[1] seasonal allergic conjunctivitis, uveitis,[2] as well as chronic forms of keratitis (e.g. adenoviral and Thygeson's keratitis), vernal keratoconjunctivitis, pingueculitis, and episcleritis.[citation needed]
Contraindications
As corticosteroids are immunosuppressive, loteprednol is contraindicated in patients with viral, fungal or mycobacterial infections of the eye.[1][2][3]
Adverse effects
Common adverse effects include foreign body sensation in the eye, dry eye and epiphora (overflow of tears), chemosis (swelling of the conjunctiva), headache, and itching. Increased intraocular pressure, a side effect typical of corticosteroids, occurs in about 2% of patients[1][2] (compared to 7% under prednisolone acetate and 0.5% under placebo).[3]
Interactions
The effect of drugs lowering intraocular pressure may be reduced. Loteprednol is not detectable in the bloodstream; so interactions with systemic drugs are highly unlikely.[1]
Pharmacology
Mechanism of action
Pharmacokinetics
Neither loteprednol etabonate nor its inactive metabolites Δ1-cortienic acid and Δ1-cortienic acid etabonate are detectable in the bloodstream, even after oral administration. A study with patients receiving loteprednol eye drops over 42 days showed no adrenal suppression, which would be a sign of the drug reaching the bloodstream to a clinically relevant extent.[1]
Steroid receptor affinity was 4.3 times that of dexamethasone in animal studies.[1]
Retrometabolic drug design
Loteprednol etabonate was developed using retrometabolic drug design. It is a so-called soft drug, meaning its structure was designed so that it is predictably metabolised to inactive substances. These metabolites, Δ1-cortienic acid and its etabonate, are derivatives of cortienic acid, itself an inactive metabolite of hydrocortisone.[1][3][4]
-
Δ1-Cortienic acid, inactive metabolite of loteprednol
-
Cortienic acid, inactive metabolite of hydrocortisone
-
Hydrocortisone, the "parent compound" of corticosteroids
Chemistry
Loteprednol etabonate is an ester of loteprednol with etabonate (ethyl carbonate), with a melting point between 220.5 °C (428.9 °F) and 223.5 °C (434.3 °F). Its solubility in water is 1:2,000,000.[3] The ketone in the side chain of classical corticosteroids such as hydrocortisone is replaced by a cleavable ester, which accounts for the rapid inactivation.[5] (This is not the same as the etabonate ester.)
Chemical synthesis
This section needs expansion. You can help by adding to it. (June 2016) |
References
- ^ a b c d e f g h Haberfeld, H., ed. (2015). Austria-Codex (in German). Vienna: Österreichischer Apothekerverlag.
- ^ a b c Loteprednol Professional Drug Facts.
- ^ a b c d Dinnendahl, V.; Fricke, U., eds. (2008). Arzneistoff-Profile (in German). Vol. 6 (22 ed.). Eschborn, Germany: Govi Pharmazeutischer Verlag. ISBN 978-3-7741-9846-3.
- ^ Bodor, N.; Buchwald, P. (2002). "Design and development of a soft corticosteroid, loteprednol etabonate". In Schleimer, R.P.; O'Byrne, P.M.; Szefler, S.J.; Brattsand, R. (eds.). Inhaled Steroids in Asthma. Optimizing Effects in the Airways. Marcel Dekker, New York. pp. 541–564.
{{cite book}}:|work=ignored (help) - ^ Pavesio, C.E.; Decory, H.H. (2008). "Treatment of ocular inflammatory conditions with loteprednol etabonate". Br J Ophthalmol. 92 (4): 455–459. doi:10.1136/bjo.2007.132621. PMID 18245274.
- ^ Druzgala, P.; Hochhaus, G.; Bodor, N. (1991). "Soft drugs—10. Blanching activity and receptor binding affinity of a new type of glucocorticoid: Loteprednol etabonate". J. Steroid Biochem. Mol. Biol. 38 (2): 149–54. doi:10.1016/0960-0760(91)90120-T. PMID 2004037.
{{cite journal}}: CS1 maint: multiple names: authors list (link)