Cadmium iodide

| |
| |

| |
| Names | |
|---|---|
| IUPAC name
Cadmium(II) iodide
| |
| Other names
Cadmium diiodide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.029.294 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| CdI2 | |
| Molar mass | 366.22 g/mol |
| Appearance | white to pale yellow crystals |
| Density | 5.640 g/cm3, solid |
| Melting point | 387 °C (729 °F; 660 K) |
| Boiling point | 742 °C (1,368 °F; 1,015 K) |
| 787 g/L (0 °C) 847 g/L (20 °C) 1250 g/L (100 °C) | |
| Solubility | soluble in ethanol, acetone, ether and ammonia |
| -117.2·10−6 cm3/mol | |
| Structure | |
| Trigonal, hP3, space group P3m1, No. 164 | |
| octahedral | |
| Hazards | |
| GHS labelling: | |
  
| |
| Danger | |
| H301, H331, H351, H373, H410 | |
| P260, P280, P301+P330+P331, P304+P340, P310, P311, P403+P233 | |
| NFPA 704 (fire diamond) | |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
[1910.1027] TWA 0.005 mg/m3 (as Cd)[1] |
REL (Recommended)
|
Ca[1] |
IDLH (Immediate danger)
|
Ca [9 mg/m3 (as Cd)][1] |
| Related compounds | |
Other anions
|
cadmium fluoride cadmium chloride cadmium bromide |
Other cations
|
zinc iodide mercury(II) iodide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cadmium iodide is an inorganic compound with the formula CdI2. It is a white hygroscopic solid. It also can be obtained as a mono- and tetrahydrate.[2] It has few applications. It is notable for its crystal structure, which is typical for compounds of the form MX2 with strong polarization effects.
Preparation
Cadmium iodide is prepared by the addition of cadmium metal, or its oxide, hydroxide or carbonate to hydroiodic acid. Also, the compound can be made by heating cadmium with iodine.[2]
Applications
Historically, cadmium iodide was used as a catalyst for the Henkel process, a high-temperature isomerisation of dipotassium phthalate to yield the terephthalate. The salt was then treated with acetic acid to yield potassium acetate and commercially valuable terephthalic acid.[3]
While uneconomical compared to the production of terephthalic acid from p-xylene, the Henkel method has been proposed as a potential route to produce terephthalic acid from furfural. As existing Bio-PET is still reliant on petroleum as a source of p-xylene, the Henkel process could theoretically offer a completely bioplastic route to polyethylene terephthalate.[4]
Crystal structure
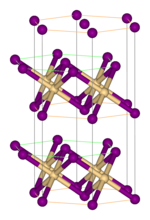
In cadmium iodide the iodide anions form a hexagonal closely packed arrangement while the cadmium cations fill all of the octahedral sites in alternate layers. The resultant structure consists of a layered lattice. This same basic structure is found in many other salts and minerals. Cadmium iodide is mostly ionically bonded but with partial covalent character.[5]
Cadmium iodide's crystal structure is the prototype on which the crystal structures of many other compounds can be considered to be based. Compounds with any of the following characteristics tend to adopt the CdI2 structure:[citation needed]
- Iodides of moderately polarising cations; bromides and chlorides of strongly polarising cations
- Hydroxides of dications, i.e. compounds with the general formula M(OH)2
- Sulfides, selenides and tellurides (chalcogenides) of tetracations, i.e. compounds with the general formula MX2, where X = S, Se, Te
References
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0087". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b F. Wagenknecht; R. Juza (1963). "Cadmium iodide". In G. Brauer (ed.). Handbook of Preparative Inorganic Chemistry, 2nd Ed. Vol. 2. NY, NY: Academic Press. p. 1096.
- ^ Solomon, I.; Eisenberg, W. (1976). Utilization of coal conversion process by-products. Quarterly report, December 27, 1975--March 27, 1976 (Report). doi:10.2172/7186862.
- ^ Tachibana, Yuya; Kimura, Saori; Kasuya, Ken-Ichi (2015). "Synthesis and Verification of Biobased Terephthalic Acid from Furfural". Scientific Reports. 5: 8249. Bibcode:2015NatSR...5E8249T. doi:10.1038/srep08249. PMC 4316194. PMID 25648201.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. pp. 1211–1212. ISBN 978-0-08-037941-8.

