Permethrin
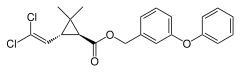
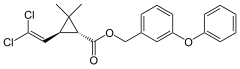
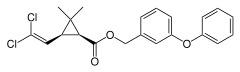
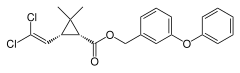
 Two-dimensional chemical structure of permethrin | |
 Three-dimensional chemical structure of permethrin | |
| Clinical data | |
|---|---|
| Trade names | Nix, Rid, Elimite, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a698037 |
| License data |
|
| Routes of administration | Topical |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Metabolism | Insects are more affected by permethrin than humans or dogs because they are unable to metabolise the toxins as quickly as humans and dogs. Cats are more sensitive to this toxin.[4] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.052.771 |
| Chemical and physical data | |
| Formula | C21H20Cl2O3 |
| Molar mass | 391.29 g·mol−1 |
| 3D model (JSmol) | |
| Density | 1.19 g/cm3, solid g/cm3 |
| Melting point | 34 °C (93 °F) |
| Boiling point | 200 °C (392 °F) |
| Solubility in water | 5.5 x 10−3 ppm, 0.2 [5] mg/mL (25°C) |
| |
| |
Permethrin is a medication and an insecticide.[6][7] As a medication, it is used to treat scabies and lice.[8] It is applied to the skin as a cream or lotion.[6] As an insecticide, it can be sprayed onto outer clothing or mosquito nets to kill the insects that touch them.[7][9]
Side effects include rash and irritation where it is applied.[8] Use during pregnancy appears to be safe.[6] It is approved for use on and around people over the age of two months.[6] Permethrin is in the pyrethroid family of medications.[6] It works by disrupting the function of the neurons of lice and scabies mites.[6]
Permethrin was discovered in 1972.[10] It is on the World Health Organization's List of Essential Medicines.[11] In 2022, it was the 351st most commonly prescribed medication in the United States, with more than 40,000 prescriptions.[12]
Uses
[edit]Insecticide
[edit]- In agriculture, to protect crops (a drawback is that it is lethal to bees)
- In agriculture, to kill livestock parasites
- For industrial and domestic insect control
- In the textile industry, to prevent insect attack of woollen products
- In aviation, the WHO, IHR and ICAO require arriving aircraft be disinsected prior to embarkation, departure, descent, or deplaning in certain countries. Aircraft disinsection with permethrin-based products is recommended only prior to embarkation. Prior to departure (after boarding), at the top of descent or on arrival, d-phenothrin-based (1R-trans phenothrin) aircraft insecticides are recommended.

Insect incapacitation
[edit]- As a personal protective measure, 0.5% permethrin is applied to outer clothing. It is a cloth impregnant, notably in mosquito nets and field wear. While permethrin may be marketed as an insect repellent, it does not prevent insects from landing. Instead it works by incapacitating or killing insects on contact before they can bite. In 2016, Consumer Reports found that, as consecutive washes reduce permethrin concentration, incapacitation becomes too slow to prevent bites. In these cases, other common topical repellents such as icaridin may be applied to the clothing, though some, such as DEET and IR3535, can damage certain synthetic fabrics.[13][9][14][15]
- In pet flea preventive collars or treatment (safe for use on dogs but not cats[16])
- In timber treatment
Medical use
[edit]This article is written like a manual or guide. (February 2024) |
Permethrin is available for topical use as a cream or lotion. It is indicated for the treatment and prevention in exposed individuals of head lice and treatment of scabies.[17]
For treatment of scabies: Adults and children older than 2 months are instructed to apply the cream to the entire body from head to the soles of the feet. Wash off the cream after 8–14 hours. In general, one treatment is curative.[18] A single application of permethrin is more effective than a single oral dose of ivermectin for scabies. In addition permethrin provides more rapid symptomatic relief than ivermectin.[19] When a second dose of ivermectin is days later, the efficacy between permethrin and ivermectin approach parity.[20]
For treatment of head lice: Apply to hair, scalp, and neck after shampooing. Leave in for 10 minutes and rinse. Avoid contact with eyes.[21]
Mechanism of action
[edit]Permethrin acts on the nerve cell membrane to disrupt the sodium channel current by which the polarization of the membrane is regulated. Delayed repolarization and paralysis of the pests are the consequences of this disturbance.[22][23]
Pest control / effectiveness and persistence
[edit]In agriculture, permethrin is mainly used on cotton, wheat, maize, and alfalfa crops. Its use is controversial because, as a broad-spectrum chemical, it kills indiscriminately; as well as the intended pests, it can harm beneficial insects, including honey bees, as well as cats and aquatic life.[24][25]
Permethrin kills ticks and mosquitoes on contact with treated clothing. A method of reducing deer tick populations by treating rodent vectors involves stuffing biodegradable cardboard tubes with permethrin-treated cotton. Mice collect the cotton for lining their nests. Permethrin on the cotton kills any immature ticks feeding on the mice.[citation needed]
Permethrin is used in tropical areas to prevent mosquito-borne disease such as dengue fever and malaria. Mosquito nets used to cover beds may be treated with a solution of permethrin. This increases the effectiveness of the bed net by killing parasitic insects before they are able to find gaps or holes in the net. Personnel working in malaria-endemic areas may be instructed to treat their clothing with permethrin as well.[citation needed]
Permethrin is the most commonly used insecticide worldwide for the protection of wool from keratinophagous insects such as Tineola bisselliella.[26]
To better protect soldiers from the risk and annoyance of biting insects, the British[27] and US armies are treating all new uniforms with permethrin.[28]
Permethrin (as well as other long-term pyrethroids) is effective over several months, in particular when used indoors. International studies report that permethrin can be detected in house dust, in fine dust, and on indoor surfaces even years after the application. Its degradation rate under indoor conditions is approximately 10% after 3 months.[29][30]
Resistance
[edit]In Aedes aegypti permethrin resistance is via "knockdown resistance" (kdr) mutations which is common to pyrethroids and DDT. This differs to the most common mechanism of insecticide resistance evolution which is selection for preexisting, low-frequency alleles. García et al. 2009 found that a kdr allele has rapidly spread throughout Mexico and become dominant there.[31]
Adverse effects
[edit]Permethrin is moderately toxic if ingested, causing abdominal pain, sore throat, nausea and vomiting. If inhaled, permethrin may cause headache, respiratory irritation, difficulty breathing, dizziness, nausea and vomiting. Inhalation is more likely from aerosols than from vapors from surfaces and clothing, as permethrin has a low vapor pressure and volatilizes slowly.[32]
Topical application of permethrin can cause mild skin irritation, burning and paresthesia.[32] Permethrin has little systemic absorption, and is considered safe for topical use in adults and children over the age of two months. The FDA has assigned it as pregnancy category B. Animal studies have suggested that it may cause endocrine disruption by interfering with estrogenic activity[32] and have shown no effects on fertility or teratogenicity, but studies in humans have not been performed. The excretion of permethrin in breastmilk is unknown, and it is recommended that breastfeeding be temporarily discontinued during treatment.[21] Skin reactions are uncommon.[33] Excessive exposure to permethrin can cause nausea, headache, muscle weakness, excessive salivation, shortness of breath, and seizures. Worker exposure to the chemical can be monitored by measurement of the urinary metabolites, while severe overdose may be confirmed by measurement of permethrin in serum or blood plasma.[34]
Permethrin does not present any notable genotoxicity or immunotoxicity in humans and farm animals, but is classified by the EPA as a likely human carcinogen when ingested, based on reproducible studies in which mice fed permethrin developed liver and lung tumors.[35] A 2018 review failed to link permethrin exposure in humans to cancer.[36]
Pharmacokinetics
[edit]Permethrin is a chemical categorized in the pyrethroid insecticide group.[4] The chemicals in the pyrethroid family are created to emulate the chemicals found in the chrysanthemum flower.[4]
Absorption
[edit]Absorption of topical permethrin is minimal. One in vivo study demonstrated 0.5% absorption in the first 48 hours based upon excretion of urinary metabolites.[37]
Distribution
[edit]Distribution of permethrin has been studied in rat models, with highest amounts accumulating in fat and the brain.[38] This can be explained by the lipophilic nature of the permethrin molecule.[citation needed]
Metabolism
[edit]Metabolism of permethrin occurs mainly in the liver, where the molecule undergoes oxidation by the cytochrome P450 system, as well as hydrolysis, into non-toxic metabolites.[37]
Excretion
[edit]The elimination of permethrin and its metabolites occurs mainly through urinary excretion, but also through feces. In rats, the excretion half-life is 12 hours for plasma and 9 to 23 hours for certain nervous tissue.[32]
Stereochemistry
[edit]Permethrin has four stereoisomers (two enantiomeric pairs), arising from the two stereocenters in the cyclopropane ring. The trans enantiomeric pair is known as transpermethrin. (1R,3S)-trans and (1R,3R)-cis enantiomers are responsible for the insecticidal properties of permethrin.[39]
-
(1S,3R)-trans enantiomer
-
(1R,3S)-trans enantiomer
-
(1S,3S)-cis enantiomer
-
(1R,3R)-cis enantiomer
Environment
[edit]Permethrin has a half-life of about 40 days in soil, 1–3 weeks on the surface of plants, over 20 days indoors, and 19–27 hours in the water column.[40] Permethrin-contaminated indoor surfaces can be decontaminated with bleach.[41]
History
[edit]In the early 1970s, it was identified that in many pyrethroids, including all natural pyrethrins and some synthetic analogs developed by that time (such as resmethrin), the furan ring, being a probable site for photo-sensitized attack by oxygen, was responsible for their instability in air and light. Hence, a group of agricultural chemists at the Rothamsted Experimental Station led by Michael Elliott tried to substitute the 5-benzyl-3-furylmethyl alcohol with quite a few structurally similar ones. Discovering that an ester of 3-phenoxybenzyl alcohol with a slightly modified (chlorine-substituted) analog of the chrysanthemic acid they also found earlier was both photo-stable and very toxic for insects, they filed their patent applications in 1972 and published their results in Nature in 1973.[10][42]
Numerous synthetic routes exist for the production of the DV-acid ester precursor.[43] The pathway known as the Kuraray Process uses four steps.[44] In general, the final step in the total synthesis of any of the synthetic pyrethroids is a coupling of a DV-acid ester and an alcohol. In the case of permethrin synthesis, the DV-acid cyclopropanecarboxylic acid, 3-(2,2-dichloroethenyl)-2,2-dimethyl-, ethyl ester, is coupled with the alcohol, m-phenoxybenzyl alcohol, through a transesterification reaction with base. Tetraisopropyl titanate or sodium ethylate may be used as the base.[44]
The alcohol precursor may be prepared in three steps. First, m-cresol, chlorobenzene, sodium hydroxide, potassium hydroxide, and cuprous chloride react to yield m-phenoxytoluene.[45] Second, oxidation of m-phenoxytoluene over selenium dioxide provides m-phenoxybenzaldehyde. Third, a Cannizzaro reaction of the benzaldehyde in formaldehyde and potassium hydroxide affords the m-phenoxybenzyl alcohol.[44]
Brand names
[edit]In Nordic countries and North America, a permethrin formulation for lice treatment is marketed under trade name Nix, available over the counter. Johnson & Johnson's UK brand Lyclear covers an assortment of different products, mostly non-insecticidal, but a few of which are based on permethrin.[46]
Stronger concentrations of permethrin are used to treat scabies (which embed inside the skin), compared to lice (which remain outside the skin). In the U.S. the more concentrated products such as Elimite are available by prescription only.[3][47]
Other animals
[edit]It is known to be highly toxic to cats, fish and aquatic species with long-lasting effects.[4][48]
Cats
[edit]Permethrin is toxic to cats; however, it has little effect on dogs.[4][49][50] Many cats die after being given flea treatments intended for dogs, or by contact with dogs having recently been treated with permethrin.[51] In cats it may induce hyperexcitability, tremors, seizures, and death.[52]
Toxic exposure of permethrin can cause several symptoms, including convulsion, hyperaesthesia, hyperthermia, hypersalivation, and loss of balance and coordination. Exposure to pyrethroid-derived drugs such as permethrin requires treatment by a veterinarian, otherwise the poisoning is often fatal.[53][54] This intolerance is due to a defect in glucuronosyltransferase, a common detoxification enzyme in other mammals, that also makes the cat intolerant to paracetamol (acetaminophen).[55] Based on those observations, the use of any external parasiticides based on permethrin is contraindicated for cats.
Aquatic organisms
[edit]Permethrin is listed as a "restricted use" substance by the US Environmental Protection Agency (EPA)[56] due to its high toxicity to aquatic organisms,[57] so permethrin and permethrin-contaminated water should be properly disposed of. Permethrin is quite stable, having a half life of 51–71 days in an aqueous environment exposed to light. It is also highly persistent in soil.[58]
References
[edit]- ^ "Nix Product Information". Health Canada. Retrieved 19 June 2024.
- ^ "Good Sense Lice Killing Creme Rinse- permethrin lotion". DailyMed. 21 November 2022. Retrieved 29 December 2024.
- ^ a b "Elimite- permethrin cream". DailyMed. 24 January 2024. Retrieved 29 December 2024.
- ^ a b c d e "Permethrin General Fact Sheet". npic.orst.edu. Archived from the original on 3 January 2018. Retrieved 6 January 2018.
- ^ Metcalf RL, Horowitz AR (19 November 2014). "Insect Control, 2. Individual Insecticides". Ullmann's Encyclopedia of Industrial Chemistry: 1–94. doi:10.1002/14356007.s14_s01. ISBN 978-3-527-30673-2.
- ^ a b c d e f "Permethrin". The American Society of Health-System Pharmacists. Archived from the original on 21 December 2016. Retrieved 8 December 2016.
- ^ a b Keystone JS, Kozarsky PE, Freedman DO, Connor BA (2013). Travel Medicine. Elsevier Health Sciences. p. 58. ISBN 978-1-4557-1076-8. Archived from the original on 20 December 2016.
- ^ a b World Health Organization (2009). Stuart MC, Kouimtzi M, Hill SR (eds.). WHO Model Formulary 2008. World Health Organization. p. 213. hdl:10665/44053. ISBN 978-92-4-154765-9.
- ^ a b "Repellent-Treated Clothing". United States Environmental Protection Agency. 15 July 2013. Retrieved 28 May 2023.
- ^ a b Elliott M, Farnham AW, Janes NF, Needham PH, Pulman DA, Stevenson JH (November 1973). "A photostable pyrethroid". Nature. 246 (5429): 169–170. Bibcode:1973Natur.246..169E. doi:10.1038/246169a0. PMID 4586114. S2CID 4176154.
- ^ World Health Organization (2023). The selection and use of essential medicines 2023: web annex A: World Health Organization model list of essential medicines: 23rd list (2023). Geneva: World Health Organization. hdl:10665/371090. WHO/MHP/HPS/EML/2023.02.
- ^ "Permethrin Drug Usage Statistics, United States, 2013 - 2022". ClinCalc. Retrieved 30 August 2024.
- ^ Jeneen I (26 May 2016). "Can Permethrin Treated Clothing Help You Avoid Mosquito Bites?". Consumer Reports. Archived from the original on 31 May 2019. Retrieved 31 May 2019.
- ^ "How to Use Permethrin on Clothing, Safely". Consumer Reports. Archived from the original on 6 May 2023. Retrieved 29 May 2023.
- ^ "How to Use Permethrin". U.S. Centers for Disease Control and Prevention (CDC). 18 March 2020. Retrieved 11 September 2023.
- ^ Malik R, Ward MP, Seavers A, Fawcett A, Bell E, Govendir M, et al. (January 2010). "Permethrin spot-on intoxication of cats Literature review and survey of veterinary practitioners in Australia". Journal of Feline Medicine and Surgery. 12 (1): 5–14. doi:10.1016/j.jfms.2009.12.002. PMC 10845477. PMID 20123482. S2CID 1234198.
- ^ "Permethrin (Lexi-Drugs)". Lexicomp Online. Wolters Kluwer. Archived from the original on 17 April 2014. Retrieved 19 April 2014.
- ^ "Permethrin Patient Package Insert" (PDF). FDA. Archived (PDF) from the original on 20 April 2014. Retrieved 19 April 2014.
- ^ Usha V, Gopalakrishnan Nair TV (February 2000). "A comparative study of oral ivermectin and topical permethrin cream in the treatment of scabies". Journal of the American Academy of Dermatology. 42 (2 Pt 1) (published February 2000): 236–240. doi:10.1016/S0190-9622(00)90131-2. PMID 10642678.
- ^ Dhana A, Yen H, Okhovat JP, Cho E, Keum N, Khumalo NP (January 2018). "Ivermectin versus permethrin in the treatment of scabies: A systematic review and meta-analysis of randomized controlled trials". Journal of the American Academy of Dermatology. 78 (1) (published January 2018): 194–198. doi:10.1016/j.jaad.2017.09.006. PMID 29241784. S2CID 29398956.
- ^ a b "Package Label" (PDF). Alpharma, USPD, Inc. Baltimore. Archived (PDF) from the original on 20 April 2014. Retrieved 19 April 2014.
- ^ "MarkerDB". markerdb.ca. Archived from the original on 2 January 2024. Retrieved 2 January 2024.
- ^ "Permethrin Cream, 5%*". dailymed.nlm.nih.gov. Retrieved 2 January 2024.
- ^ Ian RH (1989). "Aquatic organisms and pyrethroids". Pesticide Science. 27 (4): 429–457. doi:10.1002/ps.2780270408.
- ^ Drago B, Shah NS, Shah SH (2014). "Acute permethrin neurotoxicity: Variable presentations, high index of suspicion". Toxicology Reports. 1: 1026–1028. Bibcode:2014ToxR....1.1026D. doi:10.1016/j.toxrep.2014.09.007. PMC 5598406. PMID 28962315.
- ^ Ingham PE, McNeil SJ, Sunderland MR (2012). "Functional finishes for wool – Eco considerations". Advanced Materials Research. 441: 33–43. doi:10.4028/www.scientific.net/amr.441.33. S2CID 136607213.
- ^ "What's in the Black Bag?" (PDF). Personal Clothing – British Army Website. Archived from the original (PDF) on 23 September 2015. Retrieved 14 October 2015.
- ^ "Insect-repelling ACUs now available to all Soldiers". Canadian and United States Army. October 2012. Archived from the original on 4 October 2013.
- ^ Nakagawa LE, do Nascimento CM, Costa AR, Polatto R, Papini S (May 2020). "Persistence of indoor permethrin and estimation of dermal and non-dietary exposure". Journal of Exposure Science & Environmental Epidemiology. 30 (3): 547–553. Bibcode:2020JESEE..30..547N. doi:10.1038/s41370-019-0132-7. PMID 30926895. S2CID 85566713.
- ^ Berger-Preiess E, Preiess A, Sielaff K, Raabe M, Ilgen B, Levsen K (1997). "The Behaviour of Pyrethroids Indoors: A Model Study". Indoor Air. 7 (4): 248–262. Bibcode:1997InAir...7..248B. doi:10.1111/j.1600-0668.1997.00004.x.
- ^ Labbé P, Alout H, Djogbénou L, Pasteur N, Weill M (2011). "Evolution of Resistance to Insecticide in Disease Vectors". In Tibayrenc M (ed.). Genetics and Evolution of Infectious Disease. Elsevier. pp. 363–409. ISBN 978-0-12-384890-1.
- ^ a b c d "Permethrin". National Pesticide Information Center. Retrieved 30 August 2023.
- ^ Stafford III KC (February 1999). "Tick Bite Prevention". Connecticut Department of Public Health. Archived from the original on 12 September 2005.
- ^ Baselt R (2008). Disposition of Toxic Drugs and Chemicals in Man (8th ed.). Foster City, CA: Biomedical Publications. pp. 1215–1216.
- ^ Permethrin Facts. National Service Center for Environmental Publications (NSCEP) (Report). U.S. Environmental Protection Agency. June 2006. Archived from the original on 24 March 2023. Retrieved 24 March 2023.
- ^ Boffetta P, Desai V (July 2018). "Exposure to permethrin and cancer risk: a systematic review". Critical Reviews in Toxicology. 48 (6): 433–442. doi:10.1080/10408444.2018.1439449. PMID 29687728. S2CID 26073529.
- ^ a b van der Rhee HJ, Farquhar JA, Vermeulen NP (1989). "Efficacy and transdermal absorption of permethrin in scabies patients". Acta Dermato-Venereologica. 69 (2): 170–173. PMID 2564238.
- ^ Tornero-Velez R, Davis J, Scollon EJ, Starr JM, Setzer RW, Goldsmith MR, et al. (November 2012). "A pharmacokinetic model of cis- and trans-permethrin disposition in rats and humans with aggregate exposure application". Toxicological Sciences. 130 (1): 33–47. doi:10.1093/toxsci/kfs236. PMID 22859315. S2CID 20980566.
- ^ Dornetshuber J, Bicker W, Lämmerhofer M, Lindner W, Karwan A, Bursch W (14 November 2007). "Impact of stereochemistry on biological effects of permethrin: induction of apoptosis in human hepatoma cells (HCC-1.2) and primary rat hepatocyte cultures". BMC Pharmacology. 7 (Supplement 2): A65. doi:10.1186/1471-2210-7-S2-A65. ISSN 1471-2210.
Stereochemistry plays a crucial role in determining the toxicological profile of many chiral xenobiotics, e.g. the insecticidal action of mixtures containing the four stereoisomers of permethrin is essentially brought about by the (1R, cis)- and (1R, trans)-forms.
- ^ "Pesticide Half-life". National Pesticide Information Center. May 2015. Retrieved 11 September 2023.
- ^ Oudejans L, Mysz A, Gibb Snyder E, Wyrzykowska-Ceradini B, Nardin J, Tabor D, et al. (October 2020). "Remediating Indoor Pesticide Contamination from Improper Pest Control Treatments: Persistence and Decontamination Studies". Journal of Hazardous Materials. 397: 122743. Bibcode:2020JHzM..39722743O. doi:10.1016/j.jhazmat.2020.122743. PMC 7472880. PMID 32361138.
- ^ GB 1413491, Elliott, Michael; James, Norman Frank & Pulman, David Allen, "3-SUBSTITUTED-2,2-DIMETHYL-CYCLOPROPANE CARBOXYLIC ACID ESTERS THEIR PREPARATION AND THEIR USE IN PESTICIDAL COMPOSITIONS", published 1973-04-30, issued 1975-11-12, assigned to National Research Development Corporation
- ^ DV-acid = 3-(2,2-Dichlorovinyl)-2,2-dimethylcyclopropanecarboxylic acid
- ^ a b c Wasselle LA (June 1981). "Pyrethroid Insecticides" (PDF). SRI International Report #143. Menlo Park, CA. Archived (PDF) from the original on 16 March 2024. Retrieved 17 September 2023.
- ^ US 4229594A, Colby, Thomas H., "Preparation of meta-phenoxytoluene", published 1980-10-21
- ^ "Head Lice Treatments". Lyclear UK. Archived from the original on 25 September 2018. Retrieved 24 September 2018.
- ^ "Resources for Health Professionals: Scabies Medications". U.S. Centers for Disease Control and Prevention (CDC). 19 April 2019. Archived from the original on 30 April 2015. Retrieved 24 September 2018.
- ^ "Permethrin". pubchem.ncbi.nlm.nih.gov. PubChem. Archived from the original on 28 June 2020. Retrieved 28 June 2020.
- ^ Roberts C (22 October 2018). "Should You Use Natural Tick Prevention for Your Dog or Cat?". Consumer Reports. Consumer Reports Inc. Archived from the original on 15 December 2018. Retrieved 12 December 2018.
- ^ Englar RE (2019). Common Clinical Presentations in Dogs and Cats. John Wiley & Sons. p. 333. ISBN 978-1-119-41459-9. Archived from the original on 19 August 2020. Retrieved 31 May 2020.
- ^ Linnett PJ (2008). "Permethrin toxicosis in cats". Australian Veterinary Journal. 86 (1–2): 32–35. doi:10.1111/j.1751-0813.2007.00198.x. PMID 18271821.
- ^ Page SW (2008). "10: Antiparasitic drugs". In Maddison JE (ed.). Small Animal Clinical Pharmacology. Stephen W. Page, David Church. Elsevier Health Sciences. p. 236. ISBN 978-0-7020-2858-8. Archived from the original on 24 March 2017.
- ^ Richardson JA (June 2000). "Permethrin spot-on toxicoses in cats". Journal of Veterinary Emergency and Critical Care. 10 (2): 103–106. doi:10.1111/j.1476-4431.2000.tb00006.x.
- ^ Dymond NL, Swift IM (June 2008). "Permethrin toxicity in cats: a retrospective study of 20 cases". Australian Veterinary Journal. 86 (6): 219–223. doi:10.1111/j.1751-0813.2008.00298.x. PMID 18498556.
- ^ Bordeau W (January 2009). "Intoxications du chat par la perméthrine: rares mais graves" (PDF). Sciences & Pratique. 1018. Archived from the original (PDF) on 27 December 2013. Retrieved 26 January 2015.
- ^ "Restricted Use Products (RUP) Report". U.S. Environmental Protection Agency (EPA). 16 February 2024. Archived from the original on 11 January 2010. Retrieved 1 December 2009.
- ^ Environmental Protection Agency (June 2006). "Permethrin Facts (Reregistration Eligibility Decision (RED) Fact Sheet)" (PDF). epa.gov. EPA Special Review and Reregistration Division. Archived (PDF) from the original on 6 August 2020. Retrieved 13 May 2020.
- ^ Imgrund H (28 January 2003). "Environmental Fate of Permethrin" (PDF). Environmental Monitoring Branch, California Department of Pesticide Regulation, California Environmental Protection Agency. Archived (PDF) from the original on 7 October 2013.
External links
[edit]- Permethrin in the Pesticide Properties DataBase (PPDB)
- Permethrin General Fact Sheet National Pesticide Information Center
- Permethrin Technical Fact Sheet National Pesticide Information Center
- Permethrin-treated Clothing National Pesticide Information Center
- "Health Effects of Permethrin-Impregnated Army Battle-Dress Uniforms" National Research Council
- Permethrin Pesticide Information Profile Extension Toxicology Network